Submitted:
08 December 2024
Posted:
09 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Description of Randomized Controlled Trials
3.2. Analysis of Randomized Controlled Trials by Outcome Parameter Including Meta-Analysis
3.3. Description of Non-Randomized Controlled Trials
3.4. Tolerability
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Farthing, M.; Salam, M.; Lindberg, G.; Dite, P.; Khalif, I.; Salazar-Lindo, E.; Ramakrishna, B.S.; Goh, K.; Thomson, A.; Khan, A.G.; et al. World Gastroenterology Global Guidelines: Acute diarrhea in adults and children: a global perspective. Available online: http://www.worldgastroenterology.org/UserFiles/file/guidelines/acute-diarrhea-english-2012.pdf (accessed on 6.10.2016).
- Guarino, A.; Ashkenazi, S.; Gendrel, D.; lo Vecchio, A.; Sharmir, R.; Szajewska, H. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/European Society for Pediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 132–152. [Google Scholar] [CrossRef]
- Edelstein, M.; Merk, H.; Deogan, C.; Carnahan, A.; Wallensten, A. Quantifying the incidence and cost of acute gastrointestinal illness in Sweden, 2013–2014. Epidemiol. Infect. 2016, 144, 2831–2839. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.A.; Cumberland, P.; Sockett, P.N.; Wheeler, J.; Rodrigues, L.C.; Sethi, D.; Roderick, P.J.; Infectious Intestinal Disease Study, E. The study of infectious intestinal disease in England: socio-economic impact. Epidemiol. Infect. 2003, 130, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tam, C.C.; Rodrigues, L.C.; Viviani, L.; Dodds, J.P.; Evans, M.R.; Hunter, P.R.; Gray, J.J.; Letley, L.H.; Rait, G.; Tompkins, D.S.; et al. Longitudinal study of infectious intestinal disease in the UK (IID2 study): incidence in the community and presenting to general practice. Gut 2012, 61, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Lew, J.F.; Glass, R.I.; Gangarosa, R.E.; Cohen, I.P.; Bern, C.; Moe, C.L. Diarrheal deaths in the United States, 1979 through 1987: a special problem for the elderly. JAMA 1991, 265, 3280–3284. [Google Scholar] [CrossRef] [PubMed]
- Kosek, M.; Bern, C.; Guerrant, R.L. The global burden of diarrhoeal disease, as estimated from studies published between 1992 and 2000. Bull. World Health Organ. 2003, 81, 197–204. [Google Scholar] [PubMed]
- Eberlin, M.; Mück, T.; Michel, M.C. A comprehensive review of the pharmacodynamics, pharmacokinetics, and clinical effects of the neutral endopeptidase inhibitor racecadotril. Front. Pharmacol. 2012, 3, 93. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Loperamide. Available online: http://www.drugs.com/pro/loperamide.html (accessed on 26.04.2016).
- Szajewska, H.; Berni Canani, R.; Domellöf, M.; Guarino, A.; Hojsak, I.; Indrio, F.; Lo Vecchio, A.; Mihatsch, W.A.; Mosca, A.; Orel, R.; et al. Probiotics for the management of pediatric gastrointestinal disorders: position paper of the ESPGHAN special interest group on gut microbiota and modifications. J. Pediatr. Gastroenterol. Nutr. 2023, 76, 232–247. [Google Scholar] [CrossRef]
- Szajewska, H.; Guarino, A.; Hojsak, I.; Indrio, F.; Kolacek, S.; Orel, R.; Salvatore, S.; Shamir, R.; van Goudoever, J.B.; Vandenplas, Y.; et al. Use of probiotics for the management of acute gastroenteritis in children: an update. J. Pediatr. Gastroenterol. Nutr. 2020, 71, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Rodríguez-Bueno, C.P.; Abreu, Y.A.A.T.; Guarner, F.; Guno, M.J.V.; Pehlivanoğlu, E.; Perez, M. , 3rd. Bacillus clausii for gastrointestinal disorders: a narrative literature review. Adv. Ther. 2022, 39, 4854–4874. [Google Scholar] [CrossRef]
- Ghelardi, E.; Abreu, Y.A.A.T.; Marzet, C.B.; Alvarez Calatayud, G.; Perez, M., 3rd; Moschione Castro, A.P. Current Progress and Future Perspectives on the Use of Bacillus clausii. Microorganisms 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Vecchione, A.; Celandroni, F.; Mazzantini, D.; Senesi, S.; Lupetti, A.; Ghelardi, E. Compositional quality and potential gastrointestinal behavior of probiotic products commercialized in Italy. Front Med (Lausanne) 2018, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Kharwar, A.; Bazaz, M.R.; Dandekar, M.P. Quantitative and qualitative characterization of commercially available oral suspension of probiotic products containing Bacillus Clausii spores. BMC Microbiol. 2022, 22, 217. [Google Scholar] [CrossRef] [PubMed]
- Abbrescia, A.; Palese, L.; Papa, S.; Gaballo, A.; Alifano, P.; M Sardanelli, A. Antibiotic sensitivity of Bacillus clausii strains in commercial preparation. Clinical Immunology, Endocrine & Metabolic Drugs (Discontinued) 2014, 1, 102–110. [Google Scholar]
- Girlich, D.; Leclercq, R.; Naas, T.; Nordmann, P. Molecular and biochemical characterization of the chromosome-encoded class A beta-lactamase BCL-1 from Bacillus clausii. Antimicrob Agents Chemother 2007, 51, 4009–4014. [Google Scholar] [CrossRef]
- Bozdogan, B.; Galopin, S.; Leclercq, R. Characterization of a new erm-related macrolide resistance gene present in probiotic strains of Bacillus clausii. Appl Environ Microbiol 2004, 70, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Bozdogan, B.; Galopin, S.; Gerbaud, G.; Courvalin, P.; Leclercq, R. Chromosomal aadD2 encodes an aminoglycoside nucleotidyltransferase in Bacillus clausii. Antimicrob Agents Chemother 2003, 47, 1343–1346. [Google Scholar] [CrossRef] [PubMed]
- Galopin, S.; Cattoir, V.; Leclercq, R. A chromosomal chloramphenicol acetyltransferase determinant from a probiotic strain of Bacillus clausii. FEMS Microbiol Lett 2009, 296, 185–189. [Google Scholar] [CrossRef]
- Ianiro, G.; Rizzatti, G.; Plomer, M.; Lopetuso, L.; Scaldaferri, F.; Franceschi, F.; Cammarota, G.; Gasbarrini, A. Bacillus clausii for the treatment of acute diarrhea in children: a systematic review and meta-analysis of randomized controlled trials. Nutrients 2018, 10, 1074. [Google Scholar] [CrossRef]
- Centre for Reviews and Dissemination. Systematic Reviews. CRD's guidance for undertaking reviews in health care; York Publishing Services Ltd.: Layerthorpe, 2009; p. 294. [Google Scholar]
- Suurmond, R.; van Rhee, H.; Hak, T. Introduction, comparison, and validation of Meta-Essentials: A free and simple tool for meta-analysis. Res Synth Methods 2017, 8, 537–553. [Google Scholar] [CrossRef]
- McFarland, L.V.; Hecht, G.; Sanders, M.E.; Goff, D.A.; Goldstein, E.J.C.; Hill, C.; Johnson, S.; Kashi, M.R.; Kullar, R.; Marco, M.L.; et al. Recommendations to improve quality of probiotic systematic reviews with meta-analyses. JAMA Netw Open 2023, 6, e2346872. [Google Scholar] [CrossRef]
- Urtula, R.P.; Dacula, C.E. Bacillus clausii an adjunct treatment for pediatric patients with acute non-bloody diarrhea: a randomized, controlled clinical trial. Pediatria 2008, 2008 Suppl. S3, 14. [Google Scholar]
- Akter, S. Anti-diarrhoeal effects of probiotics on children. Chittagong Veterinary and Animal Science University, Chittagong, 2016.
- Lahiri, K.R. A phase III, controlled, open-label, randomized, parallel grops, multicentric, comparative study to assess the efficacy and safety of oral rehydration therapy (ORT) in combination with spores of Bacillus clausii (Enterogermina) versus ORT alone, administered for 5 days in th treatment of acute diarrhea in children; Seth G.S. Medical College: 9.7.2008 2008.
- Canani, R.B.; Cirillo, P.; Terrin, G.; Cesarano, L.; Spagnuolo, M.I.; De Vincenzo, A.; Albano, F.; Passariello, A.; De Marco, G.; Manguso, F.; et al. Probiotics for treatment of acute diarrhoea in children: randomised clinical trial of five different preparations. BMJ 2007, 335, 340. [Google Scholar] [CrossRef] [PubMed]
- Maugo, B.; Jowi, C.Y.; Murila, F.; Laving, A. Efficacy of Bacillus clausii in reducing duration of illness in acute diarrhoea in children 6-59 months of age ademitted with severe dehydration. East Afr. Med. J. 2021, 98, 3649–3658. [Google Scholar]
- Vidjeadevan, D.; Vinoth, S.; Ramesh, S. Role of Saccharomyces boulardii and Bacillus clausii in reducing the duration of diarrhea: a three-armed randomised controlled trial. International Journal of Contemporary Pediatrics 2018, 5, 1811–1814. [Google Scholar] [CrossRef]
- Lahiri, K.R.; Singh, R.; Apte, M.; Patil, M.; Taksande, A.; Varona, R.; Chatterjee, G.; Verma, M.; Brette, S.; Perez, M.I. Efficacy and safety of Bacillus clausii (O/C, N/R, SIN, T) probiotic combined with oral rehydration therapy (ORT) and zinc in acute diarrhea in children: a randomized, double-blind, placebo-controlled study in India. Trop Dis Travel Med Vaccines 2022, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Smiyan, O.I.; Smiian-Horbunova, K.O.; Bynda, T.P.; Loboda, A.M.; Popov, S.V.; Vysotsky, I.Y.; Moshchych, O.P.; Vasylieva, O.G.; Manko, Y.A.; Ovsianko, O.L.; et al. Optimization of the treatment of rotavirus infection in children by using bacillus clausii. Wiaddomsci Lekkarskie 2019, 72, 1320–1323. [Google Scholar] [CrossRef]
- Tkakiev, M.Y.; Krivochko, M.Y.; Chernusova, S.N.; Mikhailenko, E.P. Safe and effetive enteroprotective therapy of rotavirus infection in children. Sovremennayy Pediatriya 2013, 7, 109–112. [Google Scholar]
- Lahiri, K.; Jadhav, K.; Gahlowt, P.; Najmuddin, F. Bacillus clausii as an adjuvant therapy in acute childhood diarrhoea. ISOR Journal of Dental and Medical Sciences 2015, 14, 74–76. [Google Scholar]
- Lahiri, K.; D'Souza, J.; Gahlowt, P. Beneficial role of probiotic in acute childhood diarrhoea. Journal of Harmonized Research in Medical and Health Sciences 2015, 2, 26–30. [Google Scholar]
- Hamid, F.; Quaium, S.M.M.A.; Rahman, A. Comparative study of Bacillus clausii and multistrain probiotics in the management of acute diarrhoea in children. International Journal of Research in Medical Sciences 2019, 7, 1156–1160. [Google Scholar] [CrossRef]
- Hamid, F.; Quaium, S.M.; Rahman, A.; Huassain, T.; Tahmid Islam, T. Clinical efficacy of Bacillus clausii probiotic in the management of acute diarrhea in children. Chattogram Maa-O-Shishu Hospital Medical College Journal 2019, 18, 14–17. [Google Scholar] [CrossRef]
- Bhat, S.; Shreekrishna, G.N.; Savio, C.D. Efficacy of probiotics in acute diarrhoea in children. International Journal of Contemporary Pediatrics 2018, 5, 1646–1650. [Google Scholar] [CrossRef]
- Greuter, T.; Michel, M.C.; Thomann, D.; Weigmann, H.; Vavricka, S.R. Randomized, placebo-controlled, double-blind and open-label studies in the treatment and prevention of acute diarrhea with Enterococcus faecium SF68. Front Med 2020, 7. [Google Scholar] [CrossRef] [PubMed]
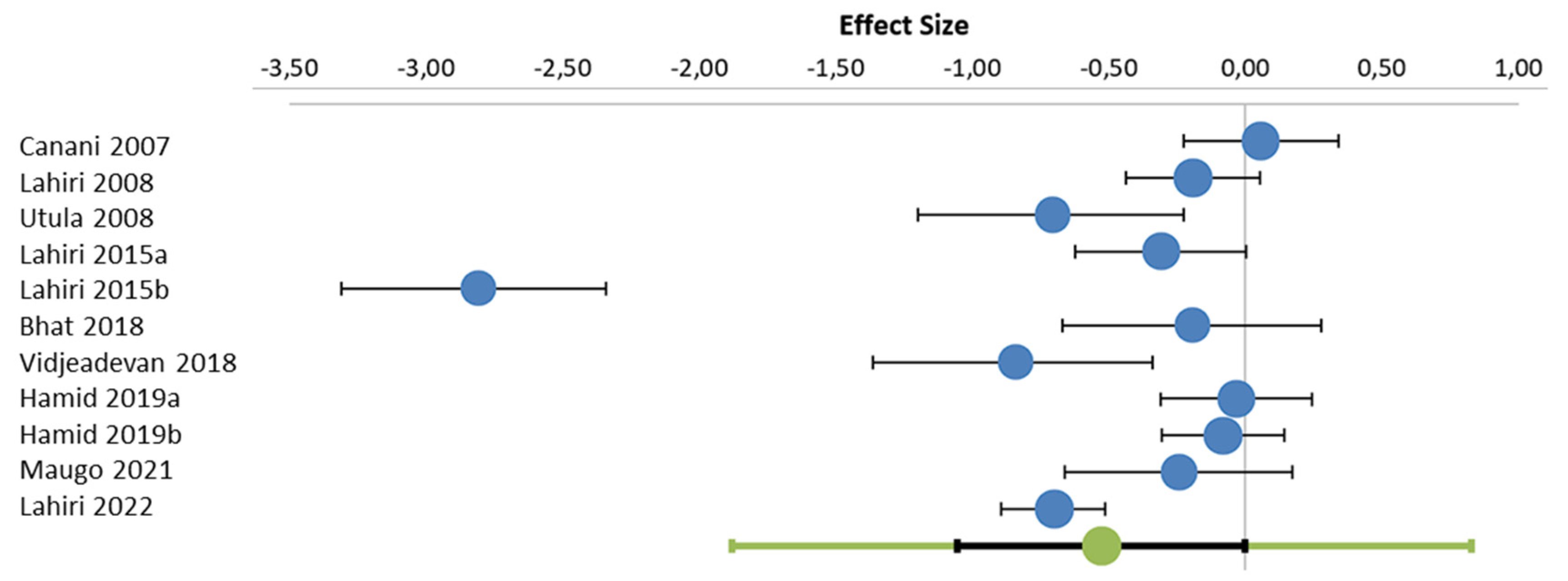
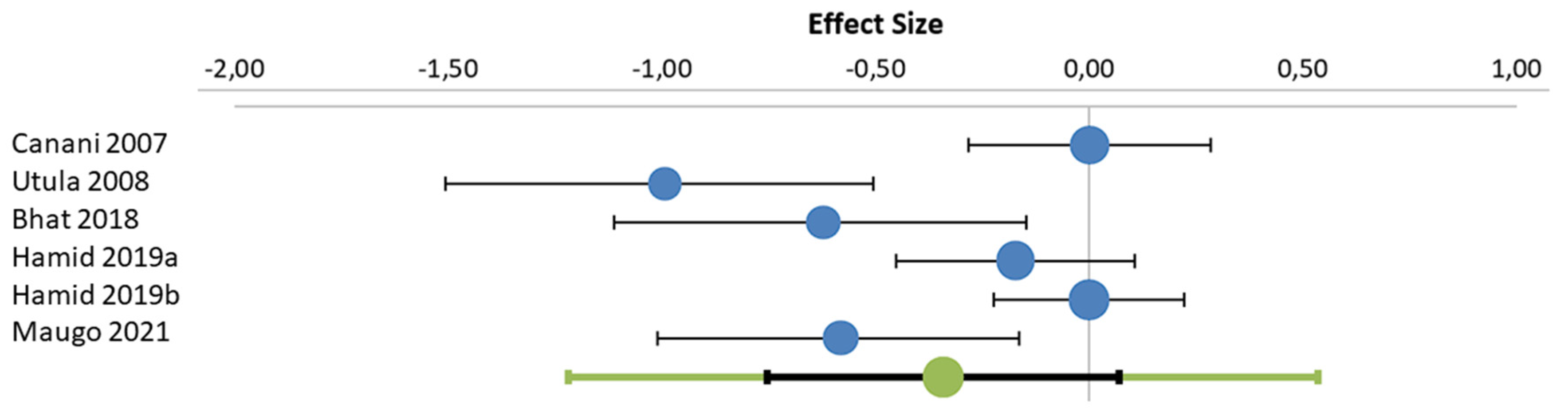
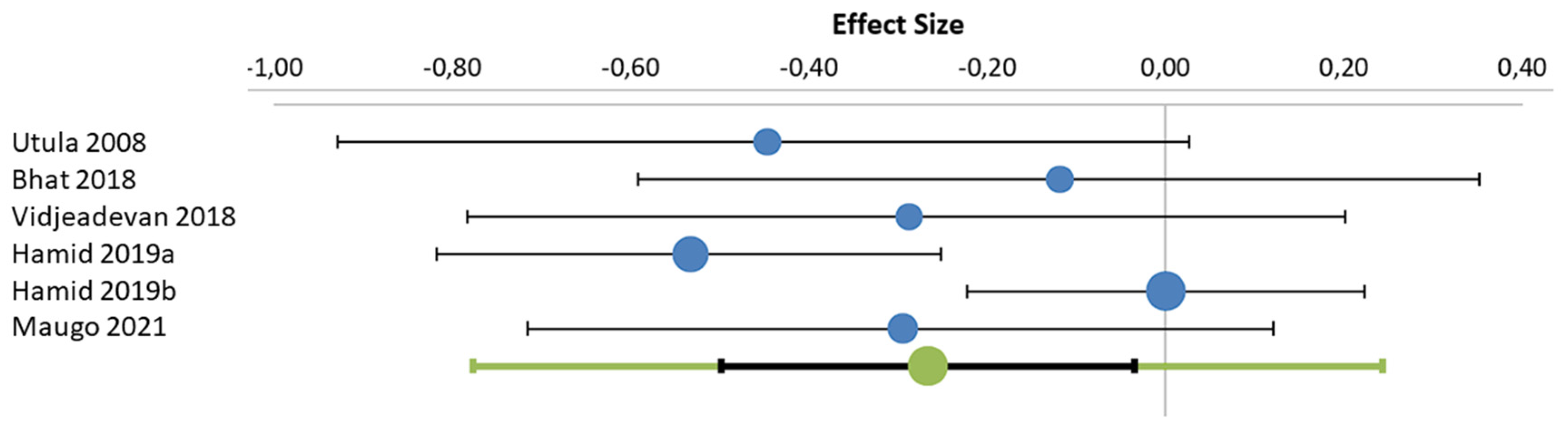
| Study | Country | Setting | Age | Comparator | Duration comparator, h |
Sample size | Total daily EG dose | Duration EG, h |
Risk of bias | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Randomized controlled trials | |||||||||||||||||||
| Canini 1997 | Italy | OP, PC | 1.6 (0.25-3) | ORT* | 112.6±23.8 | 92 vs.1080 | 2 | 114.0±25.2 | Good | ||||||||||
| Lahiri 2008 | India | OP, TCH | 1.6 ± 1.0 | ORT+zinc | 56.1±40.0 | 126 vs. 129 | 4 | 48.6±38.2 | Fair | ||||||||||
| Urtula 2008 | Philippines | IP/OP, TCH | 3.4 ± 3.3 | ORT | 83.8±22.1 | 35 vs. 35 | 2-4 | 69.8±16.8 | Fair | ||||||||||
| Lahiri 2015a | India | IP, TCH | 1.3 (0.5-6) | ORT+zinc | 34.2±1.59 | 80 vs. 80 | 4 | 22.3±1.59 | Fair | ||||||||||
| Lahiri 2015b | India | IP, TCH | n.r. (2-12) | ORT+zinc | 47.1±8.6 | 62 vs. 69 | 4 | 22.6±8.6 | Fair | ||||||||||
| Bhat 2018 | India | IP, TCH | 1.3 ± 0.6 | ORT+zinc* | 57.7±26.3 | 35 vs. 35 | 4 | 53.3±16.8 | Fair | ||||||||||
| Vidjeadevan 2018 | India | IP, TCH | 1.5±0.8 | ORT+zinc* | 108.0±8.9 | 30 vs. 30 | n.r. | 96.0±17.8 | Good | ||||||||||
| Hamid 2019a | Bangladesh | IP, TCH | 1.4±0.7 | ORT+zinc | 79.2±26.4 | 92 vs. 100 | 4 | 76.8±31.2 | Fair | ||||||||||
| Hamid 2019b | Bangladesh | IP, TCH | 1.4±0.7 | ORT+zinc* | 78.2±26.4 | 100 vs. 100 | 4 | 77.3±31.2 | Fair | ||||||||||
| Maugo 2021 | Kenya | IP, TCH | 0.9±0.4 | ORT+zinc | 86.7±40.2 | 46 vs. 44 | 4 | 77.6±34.1 | Good | ||||||||||
| Lahiri 2022 | India | IP, TCH | 2.0 (0.5-5) | ORT* | 42.8±1.0 | 227 vs. 227 | 4 | 42.1±1.0 | Good | ||||||||||
| Non-randomized controlled trials | |||||||||||||||||||
| Akter 2016 | Pakistan | IP, TCH | 1.3 (0.5-5) | ORT | 117.5±18.0 | 40 vs. 40 | 2 | 83.6±26.0 | Poor | ||||||||||
| Tkakiev 2013 | Ukraine | IP, TCH | n.r. | Other probiotic | 117.5±18.0 | 40 vs. 40 | 2-4 | 83.6±26.0 | Poor | ||||||||||
| Smiyan 2019 | Ukraine |
n.r., hospital |
n.r. (0.5-5) |
healthy | † | 28 vs. 65 | n.r. | † | Poor | ||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




