Submitted:
03 March 2024
Posted:
04 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
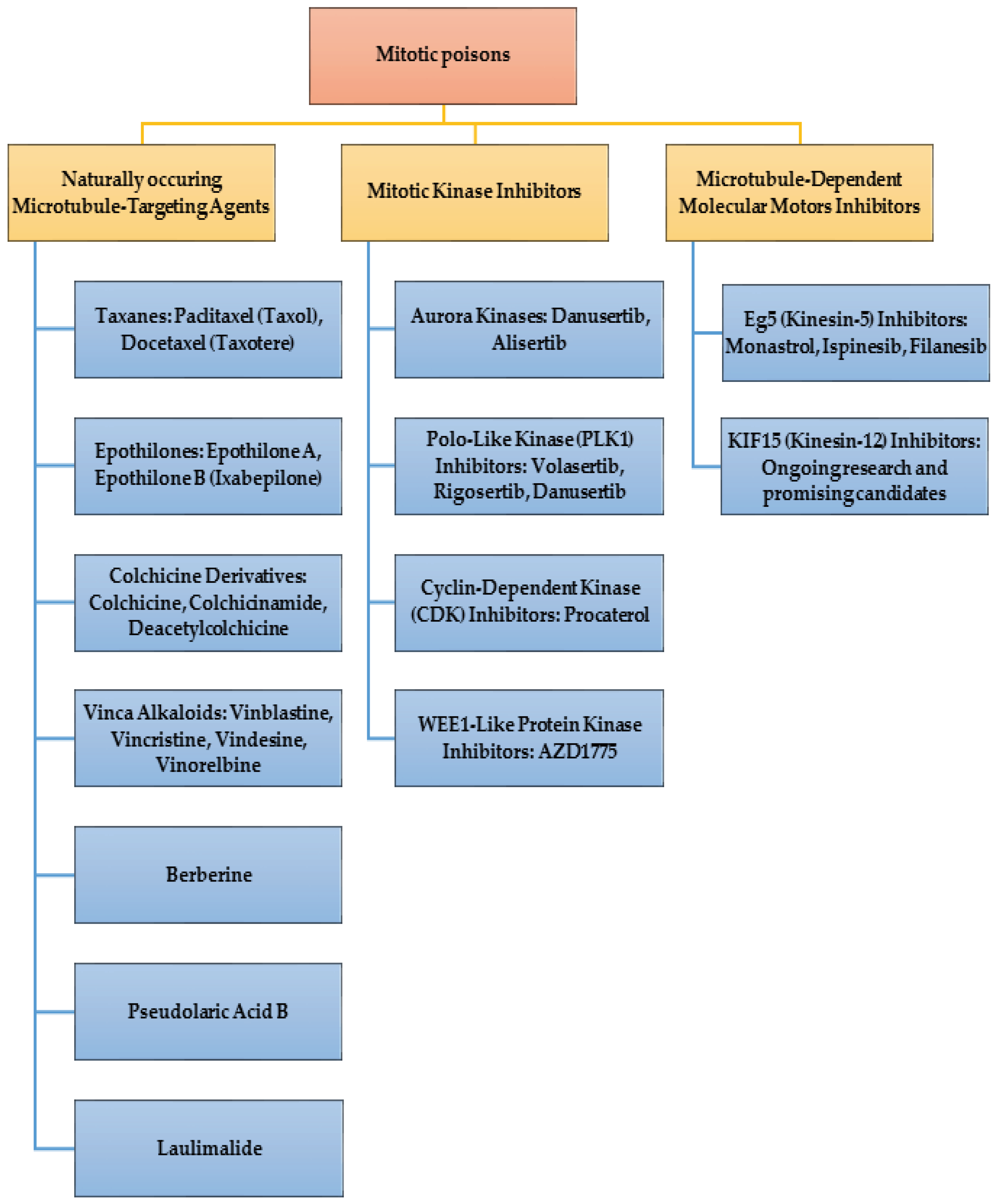
2. Mitotic Poisons
3. Targets of Mitotic Poisons
3.1. Microtubules
3.2. Mitotic Kinases
3.3. Kinesin motor proteins
4. Naturally Occurring Mitotic Poisons
4.1. Taxanes
4.2. Epothilones
4.3. Colchicine
4.4. Vinca alkaloids
4.5. Berberine
4.6. Pseudolaric Acid B
4.7. Laulimalide
5. Mitotic Kinase Inhibitors
- (i)
- Cyclin-Dependent Kinases (CDKs): are crucial regulators of the cell cycle and DNA damage response, and their overexpression is observed in various cancers, including GC [44]. A study conducted by Tang et al. unveiled that CDK2 regulates aerobic glycolysis in GC cell lines. Knocking down CDK2 led to decreased glycolytic mRNA levels and increased expression of SIRT5, a tumor suppressor involved in metabolic reprogramming [44].
- (ii)
- Aurora Kinases: are serine/threonine kinases that regulate G2/M phase transitions during cell division [45]. A study by Ding et al. revealed that Aurora B and cyclin B1 are co-expressed during the G2/M phase in GC cells. They found that Aurora B’s upregulation in the presence of high cyclin B1 levels is essential for G2/M phase transition, and it interacts with CREPT to modulate Cyclin B1 expression [46].
- (iii)
- Polo-Like Kinase 1 (PLK1): it is part of the PLK family, plays a vital role in cell cycle regulation, DNA synthesis, and p53 transactivation and is frequently overexpressed in multiple human cancers [47]. Dang et al. identified elevated PLK1 levels in GC cell lines with anti-miR-505 transfection. They demonstrated that miR-505 directly targets PLK1, thereby affecting its expression in GC cell lines [48]. Inhibition of PLK1 resulted in G2-phase cell cycle arrest and suppressed proliferation, migration, and apoptosis of GC cells. Moreover, PLK1 inhibition reduced the activation of the MEK/ERK signaling pathway [48]. Cai et al. also highlighted PLK1’s role in promoting GC cell migration, invasion, and epithelial-mesenchymal transition (EMT), indicating that PLK1 could be a potential target for therapeutic intervention in GC [49].
- (iv)
- Wee1-Like Protein Kinase (WEE1): this enzyme belongs to the Serine/Threonine protein kinase family and regulates the DNA damage checkpoint in the G2/M cell-cycle transition, enabling DNA repair before mitotic entry [50]. Wang et al. investigated WEE1’s tumorigenic role in GC and found high expression and secretion of WEE1 in GC cell lines. Silencing WEE1 through siRNA reduced cell viability in GC cell lines [51].
5.1. Mitotic Kinase Inhibitors Used against Gastric Cancers
- (i)
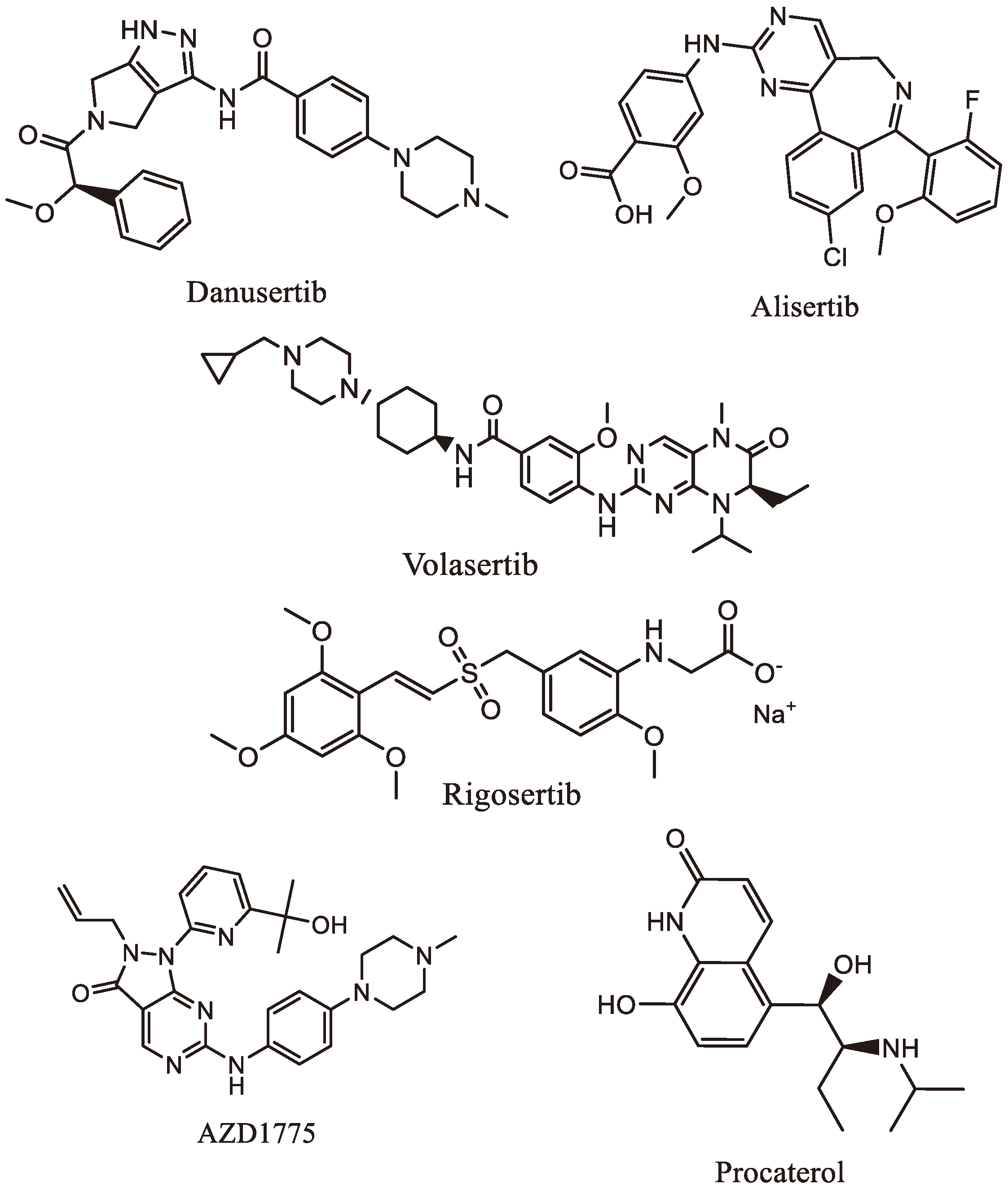
- Danusertib (Table 2 and Figure 3), previously known as PHA739358, is a potent pan-Aurora kinase inhibitor. Studies by Santo et al. indicated that Danusertib reduces the survival rate of human GC cell lines and inhibits the growth of GC cells, particularly in the G1 phase, along with reduced expression of CDK2 [52]. Kamran et al. demonstrated that Aurora A knockdown or inhibition with Alisertib (Figure 3) induced apoptosis in GC cells, reducing tumor volumes in mice [45].
- (ii)
- Volasertib (Table 2 and Figure 3), a selective PLK1 inhibitor, induces mitotic arrest and apoptosis in GC patients. Nokihara et al. confirmed the inhibitory effects of volasertib in Japanese patients with advanced GC, indicating its potential as a therapeutic option [53]. Lin et al. showed that AZD1775 (Table 2 and Figure 3), another PLK1 inhibitor, effectively targeted PLK1 in GC cells, inducing apoptosis and enhancing the inhibition of gastric tumor growth when combined with Olaparib [50].
- (iii)
- (iv)
- Liu et al. had proposed Procaterol (Table 2 and Figure 3), a β2-receptor agonist, as a potential treatment strategy for GC. They found that Procaterol suppressed cell viability and colony formation in GC cell lines and inhibited tumor growth in patient-derived gastric tumor xenografts by inhibiting CDK12 [54].
5.2. Mitotic Kinase Inhibitors Used against Liver Cancer
5.3. Mitotic Kinase Inhibitors Used against Pancreatic Cancer
5.4. Mitotic Kinase Inhibitors Used against Colorectal Cancer
6. Inhibitors of microtubule-dependent molecular motors
7. Future Perspective
Conflicts of Interest
Abbreviations
References
- Weinberg, R., Chapter 8: pRb and control of the cell cycle clock. The Biology of Cancer, 2nd Edition, 2014: p. 275-329.
- Alberts, B., et al., Molecular Biology of the Cell 6th ed (New York, NY: Garland Science). 2014.
- Hartwell, L.H. and M.B. Kastan, Cell cycle control and cancer. Science, 1994. 266(5192): p. 1821-8. [CrossRef]
- Hanahan, D. and R.A. Weinberg, Hallmarks of cancer: the next generation. Cell, 2011. 144(5): p. 646-74. [CrossRef]
- Maiato, H. and E. Logarinho, Mitotic spindle multipolarity without centrosome amplification. Nature Cell Biology, 2014. 16(5): p. 386-94. [CrossRef]
- Dumont, S. and T.J. Mitchison, Force and length in the mitotic spindle. Current Biology, 2009. 19(17): p. R749-61. [CrossRef]
- Musacchio, A. and E.D. Salmon, The spindle-assembly checkpoint in space and time. Nature reviews Molecular cell biology, 2007. 8(5): p. 379-393. [CrossRef]
- Pines, J., Mitosis: a matter of getting rid of the right protein at the right time. Trends in cell biology, 2006. 16(1): p. 55-63. [CrossRef]
- Brito, D.A. and C.L. Rieder, Mitotic checkpoint slippage in humans occurs via cyclin B destruction in the presence of an active checkpoint. Current Biology, 2006. 16(12): p. 1194-1200. [CrossRef]
- Gascoigne, K.E. and S.S. Taylor, Cancer cells display profound intra- and interline variation following prolonged exposure to antimitotic drugs. Cancer Cell, 2008. 14(2): p. 111-22. [CrossRef]
- Lara-Gonzalez, P., F.G. Westhorpe, and S.S. Taylor, The spindle assembly checkpoint. Current biology, 2012. 22(22): p. R966-R980. [CrossRef]
- Schmidt, M. and H. Bastians, Mitotic drug targets and the development of novel anti-mitotic anticancer drugs. Drug Resist Update, 2007. 10(4-5): p. 162-81. [CrossRef]
- Tischer, J. and F. Gergely, Anti-mitotic therapies in cancer. Journal of Cell Biology, 2019. 218(1): p. 10-11. [CrossRef]
- Chen, X., et al., Remote control of microtubule plus-end dynamics and function from the minus-end. Elife, 2019. 8. [CrossRef]
- Li, C., et al., Microtubule dynamic instability: the role of cracks between protofilaments. Soft Matter, 2014. 10(12): p. 2069-2080. [CrossRef]
- Strothman, C., et al., Microtubule minus-end stability is dictated by the tubulin off-rate. J Cell Biol, 2019. 218(9): p. 2841-2853. [CrossRef]
- Ferro, L.S., et al., Kinesin and dynein use distinct mechanisms to bypass obstacles. Elife, 2019. 8. [CrossRef]
- Škubník, J., et al., Mitotic poisons in research and medicine. Molecules, 2020. 25(20): p. 4632. [CrossRef]
- Jordan, M.A. and L. Wilson, Microtubules as a target for anticancer drugs. Nature reviews cancer, 2004. 4(4): p. 253-265. [CrossRef]
- Schiff, P.B. and S.B. Horwitz, Taxol stabilizes microtubules in mouse fibroblast cells. Proceedings of the National Academy of Sciences of the United States of America, 1980. 77(3): p. 1561-5. [CrossRef]
- Jordan, M.A., et al., Mechanism of mitotic block and inhibition of cell proliferation by taxol at low concentrations. Proceedings of the National Academy of Sciences of the United States of America, 1993. 90(20): p. 9552-9556. [CrossRef]
- Shi, J., J.D. Orth, and T. Mitchison, Cell type variation in responses to antimitotic drugs that target microtubules and kinesin-5. Cancer research, 2008. 68(9): p. 3269-3276. [CrossRef]
- Janssen, A., et al., Intravital FRET imaging of tumor cell viability and mitosis during chemotherapy. PloS one, 2013. 8(5). [CrossRef]
- Orth, J.D., et al., Analysis of mitosis and antimitotic drug responses in tumors by in vivo microscopy and single-cell pharmacodynamics. Cancer research, 2011. 71(13): p. 4608-4616. [CrossRef]
- Malumbres, M., Physiological relevance of cell cycle kinases. Physiological reviews, 2011. [CrossRef]
- Salmela, A.-L. and M.J. Kallio, Mitosis as an anti-cancer drug target. Chromosoma, 2013. 122: p. 431-449. [CrossRef]
- Mitchison, T., The proliferation rate paradox in antimitotic chemotherapy. Molecular biology of the cell, 2012. 23(1): p. 1-6. [CrossRef]
- Vicente, J.J. and L. Wordeman, Mitosis, microtubule dynamics and the evolution of kinesins. Experimental cell research, 2015. 334(1): p. 61. [CrossRef]
- Hirokawa, N. and Y. Tanaka, Kinesin superfamily proteins (KIFs): Various functions and their relevance for important phenomena in life and diseases. Experimental cell research, 2015. 334(1): p. 16-25. [CrossRef]
- Dehelean, C.A., et al., Plant-Derived Anticancer Compounds as New Perspectives in Drug Discovery and Alternative Therapy. Molecules, 2021. 26(4). [CrossRef]
- Nižnanský, Ľ., et al., Natural Taxanes: From Plant Composition to Human Pharmacology and Toxicity. Int J Mol Sci, 2022. 23(24). [CrossRef]
- Hao, D.C., X.-J. Gu, and P.G. Xiao, 3 - Taxus medicinal resources: a comprehensive study, in Medicinal Plants, D.C. Hao, X.-J. Gu, and P.G. Xiao, Editors. 2015, Woodhead Publishing. p. 97-136. [CrossRef]
- V Rao, C., C. D Kurkjian, and H.J.C.D.T. Y Yamada, Mitosis-targeting natural products for cancer prevention and therapy. 2012. 13(14): p. 1820-1830. [CrossRef]
- Mukhtar, E., V.M. Adhami, and H. Mukhtar, Targeting Microtubules by Natural Agents for Cancer Therapy. Molecular Cancer Therapeutics, 2014. 13(2): p. 275-284. [CrossRef]
- Ojima, I. and M. Das, Recent advances in the chemistry and biology of new generation taxoids. J Nat Prod, 2009. 72(3): p. 554-65. [CrossRef]
- Ye, W., et al., An Easy and Efficient Strategy for the Enhancement of Epothilone Production Mediated by TALE-TF and CRISPR/dcas9 Systems in Sorangium cellulosum. Front Bioeng Biotechnol, 2019. 7: p. 334. [CrossRef]
- Lau, J., et al., Optimizing the heterologous production of epothilone D in Myxococcus xanthus. Biotechnol Bioeng, 2002. 78(3): p. 280-8. [CrossRef]
- Ye, W., et al., A new approach for improving epothilone B yield in Sorangium cellulosum by the introduction of vgb epoF genes. J Ind Microbiol Biotechnol, 2016. 43(5): p. 641-50. [CrossRef]
- Dhyani, P., et al., Anticancer potential of alkaloids: a key emphasis to colchicine, vinblastine, vincristine, vindesine, vinorelbine and vincamine. Cancer Cell International, 2022. 22(1): p. 206. [CrossRef]
- Iqbal, J., et al., Plant-derived anticancer agents: A green anticancer approach. Asian Pacific Journal of Tropical Biomedicine, 2017. 7(12): p. 1129-1150. [CrossRef]
- Siddiqui, A.J., et al., Plants in Anticancer Drug Discovery: From Molecular Mechanism to Chemoprevention. BioMed Research International, 2022. 2022: p. 5425485. [CrossRef]
- Mafu, S., et al., Biosynthesis of the microtubule-destabilizing diterpene pseudolaric acid B from golden larch involves an unusual diterpene synthase. Proceedings of the National Academy of Sciences of the United States of America, 2017. 114(5): p. 974-979. [CrossRef]
- Javed, A., et al., Therapeutic Potential of Mitotic Kinases’ Inhibitors in Cancers of the Gastrointestinal System. Future Pharmacology, 2022. 2(3): p. 214-237. [CrossRef]
- Tang, Z., et al., CDK2 positively regulates aerobic glycolysis by suppressing SIRT5 in gastric cancer. Cancer Science, 2018. 109(8): p. 2590-2598. [CrossRef]
- Kamran, M., et al., Aurora kinase A regulates Survivin stability through targeting FBXL7 in gastric cancer drug resistance and prognosis. Oncogenesis, 2017. 6(2): p. e298. [CrossRef]
- Ding, L., et al., CREPT/RPRD1B associates with Aurora B to regulate Cyclin B1 expression for accelerating the G2/M transition in gastric cancer. Cell Death Dis, 2018. 9(12): p. 1172. [CrossRef]
- Wakida, T., et al., The CDK-PLK1 axis targets the DNA damage checkpoint sensor protein RAD9 to promote cell proliferation and tolerance to genotoxic stress. eLife, 2017. 6: p. e29953. [CrossRef]
- Dang, S.C., et al., MicroRNA-505 suppresses gastric cancer cell proliferation and invasion by directly targeting Polo-like kinase-1. Onco Targets Ther, 2019. 12: p. 795-803. [CrossRef]
- Cai, X.P., et al., PLK1 promotes epithelial-mesenchymal transition and metastasis of gastric carcinoma cells. Am J Transl Res, 2016. 8(10): p. 4172-4183.
- Lin, X., et al., Augmented antitumor activity by olaparib plus AZD1775 in gastric cancer through disrupting DNA damage repair pathways and DNA damage checkpoint. J Exp Clin Cancer Res, 2018. 37(1): p. 129. [CrossRef]
- Wang, C.H., et al., Tazarotene-induced gene 1 interacts with Polo-like kinase 2 and inhibits cell proliferation in HCT116 colorectal cancer cells. Cell Biol Int, 2021. 45(11): p. 2347-2356. [CrossRef]
- Santo, L., et al., AT7519, A novel small molecule multi-cyclin-dependent kinase inhibitor, induces apoptosis in multiple myeloma via GSK-3beta activation and RNA polymerase II inhibition. Oncogene, 2010. 29(16): p. 2325-36. [CrossRef]
- Nokihara, H., et al., Phase I trial of volasertib, a Polo-like kinase inhibitor, in Japanese patients with advanced solid tumors. Invest New Drugs, 2016. 34(1): p. 66-74. [CrossRef]
- Liu, H., et al., CDK12 and PAK2 as novel therapeutic targets for human gastric cancer. Theranostics, 2020. 10(14): p. 6201-6215. [CrossRef]
- Dumas, M.E., et al., Dual inhibition of Kif15 by oxindole and quinazolinedione chemical probes. Bioorganic & Medicinal Chemistry Letters, 2019. 29(2): p. 148-154. [CrossRef]
- Chandrasekaran, G., P. Tátrai, and F. Gergely, Hitting the brakes: targeting microtubule motors in cancer. British Journal of Cancer, 2015. 113(5): p. 693-698. [CrossRef]
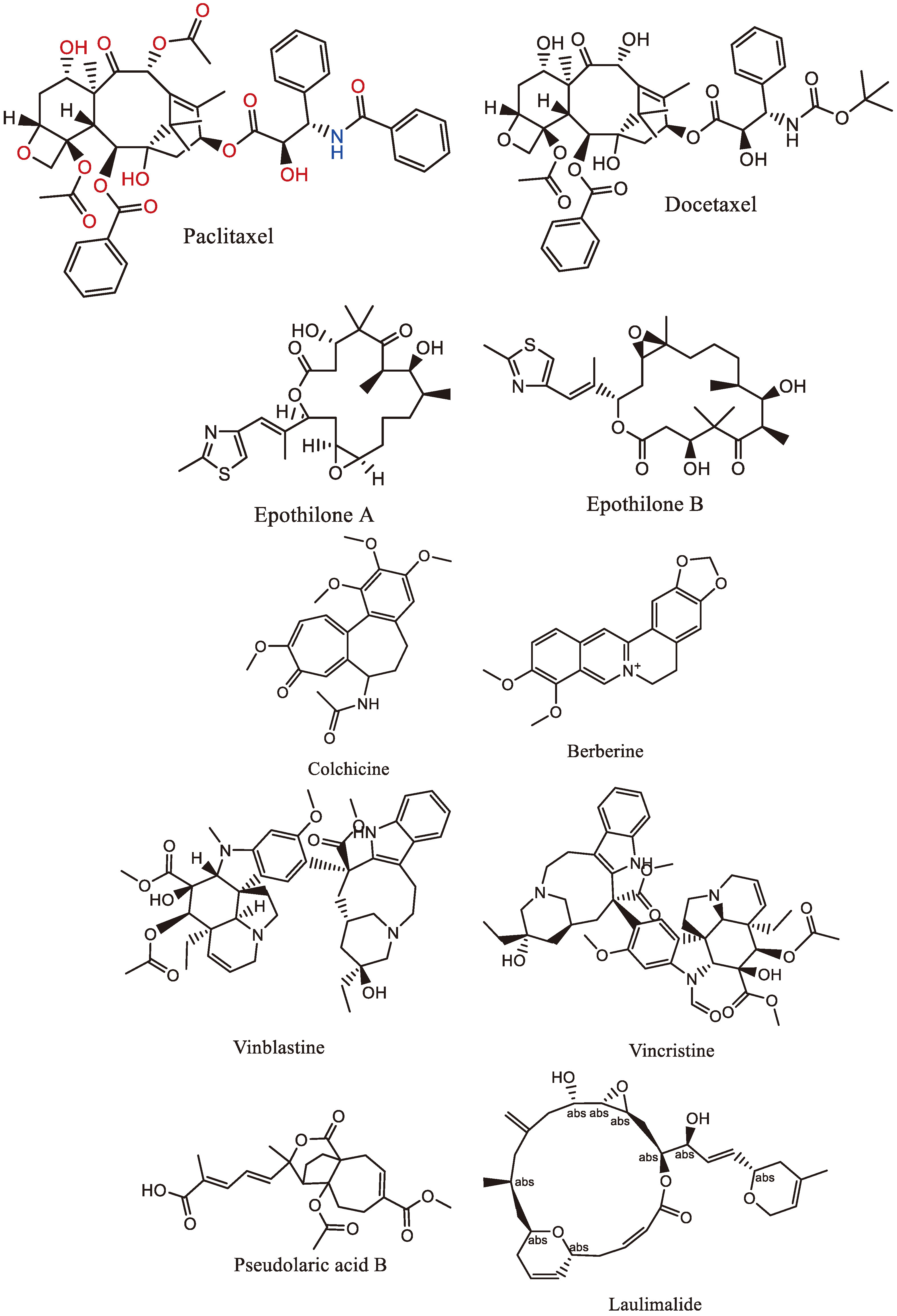
| Molecule | Chemical class | Activity/Target | Source | Organism |
|---|---|---|---|---|
| Paclitaxel | Taxanes |
They enhance microtubule polymerization. Binding to tubulin leads to the halt of cell division and triggering the activation of the mitotic spindle checkpoint. They inhibit androgen receptor signaling in prostate cancer. |
Plant | Taxus brevifolia |
| Docetaxel | Semi-synthetic | Taxus baccata | ||
| Epothilone A | Lactones | Microtubule-Targeting Agents, that stabilize microtubules, preventing cancer cells from dividing during mitosis | Bacteria | Sorangium cellulolus |
| Epothilone B | ||||
|
Vinblastine Vincristine Vindesine Vinorelbine |
Alkaloids |
They work by interfering with microtubular activity at low doses and causing cell cycle arrest and apoptosis at higher doses. | Plant | Catharanthus roseus |
| Pseudolaric acid B | Diterpene | Inhibits microtubule polymerization | Plant | Pseudolarix amabilis |
| Laulimalide | Macrolide | Inhibits microtubule polymerization | Marine sponge | Cacospongia mycofijiensis |
| Colchicine | Alkaloid | Prevents DNA synthesis and tubulin polymerization, effectively halting mitosis | Plant |
Colchicum autumnale Gloriosa superba |
| Molecule | Target/Activity | Effects |
|---|---|---|
| Danusertib | Aurora Kinase inhibitor |
1. Reduces the survival rate of human Gastric Cancer (GC) cell lines and inhibits their growth. 2. Hinders cell growth and induces cell death in pancreatic cancer. |
| Volasertib | Selective Polo-Like Kinase (PLK1) inhibitor | Induces mitotic arrest and apoptosis in GC patients. |
| AZD1775 |
Polo-Like Kinase (PLK1) inhibitor Wee1-Like Protein Kinase (WEE1) inhibitor |
1. Induces apoptosis and enhancing the inhibition of gastric tumor growth. 2. Sensitizes p53-mutated cells to chemotherapy in pancreatic cancer. |
| Procaterol |
Cyclin-Dependent Kinase (CDK12) inhibitor. β2-receptor agonist. |
Suppresses cell viability and colony formation in GC cell lines and inhibits tumor growth |
| RO-3306 | CDK1 Inhibitors |
Show promise as potential therapeutic options for liver cancer |
| Lycorine | ||
| MLN8237 | Aurora Kinase Inhibitors | |
| SNS-314 | ||
| GSK461364 | PLK1 Inhibitors | |
| Rigosertib | ||
| CCT137690 | Aurora Kinase inhibitor | Hinders cell growth and induces death in pancreatic cancer cells. |
| Dinaciclib | CDK1 Inhibitor | Induces apoptosis and immunogenic cell death in pancreatic cancer. |
| ENMD-2076 | Aurora Kinase inhibitor | Induces G2/M arrest and apoptosis in Colorectal Cancer. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




