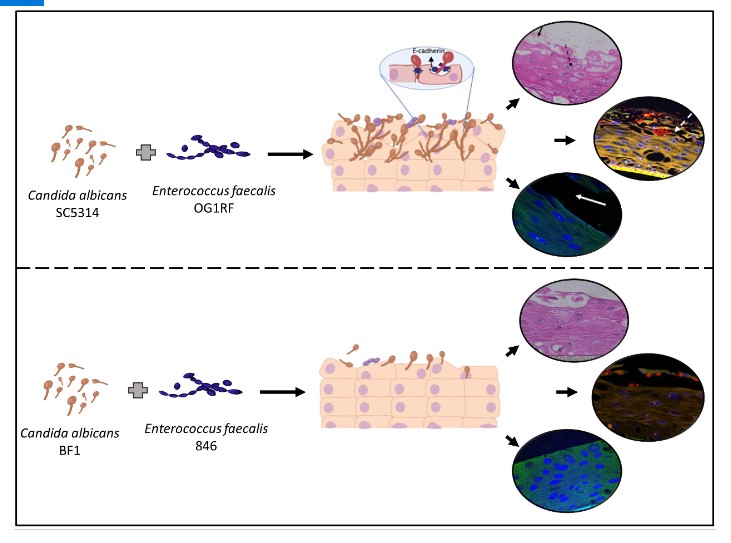
Candida albicans as an opportunistic pathogen exploits the host immune system and causes a variety of life-threatening infections. The polymorphic nature of this fungus gives it tremendous advantage to breach mucosal barriers and cause a variety of oral and disseminated infections. Enterococcus faecalis, another opportunistic pathogen co-exists with C. albicans in several niches in the human body, including the oral cavity and gastrointestinal tract. However, interactions between E. faecalis and C. albicans on oral mucosal surfaces remain unknown. Here, for the first time, we comprehensively characterized the interactive profiles between laboratory and clinical isolates of C. albicans (SC5314 and BF1) and E. faecalis (OG1RF and 846) on an organotypic oral mucosal model. Our results demonstrated that the two species formed robust biofilms on the mucosal tissue surface with profound surface erosion and fungal invasion. Specifically, this effect was more pronounced in the laboratory isolates than in the clinical isolates. Notably, several genes of C. albicans involved in tissue adhesion, hyphal formation, fungal invasion, and biofilm formation were significantly upregulated in the presence of E. faecalis. This study highlights the strain-dependent cross-kingdom interactions between E. faecalis and C. albicans on oral mucosa, demonstrating the requisite to study more substrate-dependent polymicrobial interactions.