Submitted:
05 April 2025
Posted:
08 April 2025
You are already at the latest version
Abstract
Keywords:
Index
| Chapter no. | Chapter name | Page no. |
| 1 | Introduction | 9-16 |
| 1.1 | Introduction | 9 |
| 1.2 | Aim of the Project | 9 |
| 1.3 | Risk assessment | 10 |
| 1.4 | Socio-economic importance | 10 |
| 1.5 | Scope | 11 |
| 1.6 | Leather | 11 |
| 1.7 | History of Leather Industries in Bangladesh | 13 |
| 1.8 | Tannery Industrial State | 14 |
| 1.9 | Survey Sheet | 16 |
| 2 | Production Process and Chemicals | 18-31 |
| 2.1 | General Discussion | 19 |
| 2.2 | Flow Chart of Leather Manufacturing Process | 20 |
| 2.3 | Production Management | 23 |
| 2.4 | Preparation of Wet Blue | 24 |
| 2.5 | Crusting | 27 |
| 2.6 | Finishing | 30 |
| 2.7 | Chemicals Used in the Leather Production | 31 |
| 3 | Hazardous Chemicals and Their Risk Assessments | 36-46 |
| 3.1 | General Discussion | 37 |
| 3.2 | Pictograph of Hazardous Chemicals | 38 |
| 3.3 | Highly Potential Hazard Group | 39 |
| 3.4 | Moderate Potential Hazard Group | 44 |
| 3.5 | Low Potential Hazard Group | 45 |
| 3.6 | Miscellaneous Hazard Group | 46 |
| 4 | Leather Industry and Their Effect on Environment | 47-55 |
| 4.1 | Environment | 48 |
| 4.2 | Pollution | 49 |
| 4.3 | Pollutant | 49 |
| 4.4 | Sources and Nature of Pollutant in Leather Industry | 50 |
| 4.5 | Characteristics of Tannery Discharge | 51 |
| 4.6 | Effects of Hazardous Chemicals in Water | 52 |
| 4.7 | Central Effluent Treatment Plant | 53 |
| 4.8 | Environmental Impact of The Leather Industry | 54 |
| 5 | Result and Discussion | 56-76 |
| 5.1 | Effect Description on The Basis of Interview | 57 |
| 5.2 | Univariate Analysis | 64 |
| 5.3 | Affected Workers | 70 |
| 5.4 | Impact on labor efficiency | 73 |
| 5.5 | Storage Instructions for Hazardous and Other Chemicals | 73 |
| 5.6 | Personal Protective Equipment | 75 |
| 5.7 | Work Place Criteria | 76 |
| 6 | Safety compliance of leather industry. | 78-90 |
| 6.1 | Compliance | 78 |
| 6.1.1 | Dust and fumes | 78 |
| 6.1.2 | Ventilation temperature | 79 |
| 6.1.3 | Place for child | 79 |
| 6.1.4 | Over crowding | 81 |
| 6.1.5 | Sound | 81 |
| 6.1.6 | Health care | 81 |
| 6.1.7 | First aid | 81 |
| 6.1.8 | Safety record book | 81 |
| 6.1.9 | Shelters | 82 |
| 6.2 | Maternity benefit | 84 |
| 6.3 | Worker’s responsibilities | 84 |
| 6.4 | Working hours | 85 |
| 6.5 | Use of PPE | 85 |
| 6.6 | Ergonomics | 86 |
| 6.7 | Conclusion | 88 |
| 6.8 | Recommendation | 89 |
| 6.9 | Discussion | 90 |
| References | 92-94 | |
Chapter One
- Introduction

1.1. Introduction
1.2. Aim of the Project
- To create awareness of safety and provide knowledge about chemicals among the tannery workers.
- To know about the hazardous chemicals.
- To find out the effects of the hazardous chemicals.
- To evaluate the safety measurement in the tanneries.
- To know the health problems of the workers.
- To find a better environmental measures for the workers in the tanneries.
1.3. Risk Assessment
1.4. Socio Economic Importance of the Study
1.5. Scope
1.6. Leather
- Water: 60% -70 %
- Proteins: 19.2% - 32.75%
- Fats: 1.5% - 12.25%
- Mineral Matters: 0.36% - 0.5%
- Coloring pigments: Traces
- a)
- free water
- b)
- Bound water
- Glycine 12. Histidine
- Alanine 13. Aspartic acid
- Valine 14. Glutamic acid
- Leucine 15. Tyrosine
- Serine 16. Cystine
- Tryptophan 17. Proline
- Lysine 18. Hydroxy proline
- Isoleucine 19. Arginine
- Theronine 20. Hydroxylysine
- Cysteine 21. Phenyl Alanine
- Methionine
- Pre-tanning operations
- Tanning operations
- Post-tanning operations or Finishing operations
1.7. History of Leather Industries in Bangladesh
1.8. Tannery Industrial State
- Carcinogens: Hexavalent chromium, Benzene dyes etc. are responsible for this.
- Skin disease: It includes itching, redness, rashes or swelling.
- Chemical poisoning: It means chemical irritations, allergy, vomiting, nausea etc.
- Bronchitis: It is the inflammation of the mucous membranes.
1.9. Survey Sheet
| Institute of Leather Engineering and Technology |
| University of Dhaka |
| Sl. | Question | Yes | No | Comment |
| 01 | Do you feel vomiting while working in the tannery? | |||
| 02 | Do you feel any hearing problem? | |||
| 03 | Do you have reduced eyesight? | |||
| 04 | Do you have any breathing problem? | |||
| 05 | Do you have hair fall problem? | |||
| 06 | Do you have liver problem? | |||
| 07 | Does eating food in the middle of work cause stomach pain? | |||
| 08 | Do you have sound sleep? | |||
| 09 | Has acid ever come in contact with your skin? | |||
| 10 | Ever had eye contact while handling chemicals? | |||
| 11 | Has any chemical caused any wound in your body? | |||
| 12 | Is there any allergy problem? | |||
| 13 | Do you get adequate protections during handling of hazardous chemicals? | |||
| 14 | Do you have any ideas about the harmful effects of harmful chemicals? | |||
| 15 | Is there any problem of frequent diarrhea? | |||
| Sign of surveyor Sign of the person concerned | ||||
Chapter Two
- Production Process and Chemicals

2.1. General Discussion
- Chrome-Tanned Leather is leather that has been tanned using chromium sulfate in addition to other chromium salts. This leather is hollow and has a very slight elastic quality. It survives the boiling water test. Because of this, the temperature at which it shrinks is higher than that of leather that has been vegetable-tanned. When it is sliced, the fibers work their way out into the section that has been cut. It does it in a measured manner.
- Tannins and other components are extracted from various types of vegetable matter in order to produce vegetable-tanned leather. These components can be extracted from tree bark that has been processed in bark mills, wood, leaves, fruits, and other similar sources. When compared to chrome tanned leather, this leather has a fuller appearance and less elasticity. It fails the boil test with flying colors. When it is sliced, the fibers remain in the cut section even after the piece has been removed. It has a greater capacity for absorbing fat than chrome-tanned leather.
- Leather that has been treated with aldehyde undergoes the tanning process using glutaraldehyde or oxazolidine chemicals.
2.2. Flow Chart of Leather Manufacturing Process
| Raw hides or skins selections | |
 |
|
| Sorting | |
 |
|
| Trimming | |
 |
|
| Weighing (salt weight/ green weight) | |
 |
|
| Pre-soaking | |
 |
|
| Soaking | |
 |
|
| Unhairing (if necessary) | |
 |
|
| Liming | |
 |
|
| Fleshing | |
 |
|
| Weighing (pelt weight) | |
 |
|
| Deliming | |
 |
|
| Bating | |
 |
|
| Scudding | |
 |
|
| Pickling | |
 |
|
| De-pickling (if necessary) | |
 |
|
| De-greasing (if necessary) | |
 |
|
| Pre-tanning | |
 |
|
| Basification | |
| Wet Blue Leather |  |
| Pilling | |
 |
|
| Ageing | |
 |
|
| Wet blue selection and grading | |
 |
|
| Sammying | |
 |
|
| Splitting (if necessary) | |
 |
|
| Shaving | |
 |
|
| Weighing (shaved weight) | |
 |
|
| Wet back | |
 |
|
| Re-chroming | |
 |
|
| Neutralization | |
 |
|
| Re-Tanning and Dyeing | |
 |
|
| Fatliquoring | |
 |
|
| Fixing/ Top coat | |
 |
|
| Horse up | |
 |
|
| Setting | |
 |
|
| Vacuum dry | |
 |
|
| Hang to dry | |
 |
|
| Staking | |
 |
|
| Toggling | |
 |
|
| Trimming | |
 |
|
| Kiss plate | |
| Crust Leather |  |
| Auto/Hand spraying for finishing | |
 |
|
| Roto press | |
 |
|
| Glazing/Embossing/Hair cell | |
| Finished Leather |  |
| Selection | |
 |
|
| Measurement | |
 |
|
| Packaging |
2.3. Production Management
2.4. Preparation of Wet Blue
- Curing: It is the process of keeping the leather nonputrescible for some days before the start of tanning process. This is done because of the time gap between the flaying operations and the start of the tanning process. The leather can get putrefied by the action of bacteria. Curing can be done by controlling temperature, pH, using toxic materials or by simple salting process.
- Soaking: This is the first tannery operation. The hides/skins are treated with water for the purpose of rehydration of skin proteins. The fibrous structure of the skins are opened. The surface is cleaned and curing salts, dirts, blood etc. are removed in this process. The soaking process varies according to the curing process. Soaking of dry salted hides or skins is very difficult to soak it back.
- Liming: The hides and skins are taken for liming operation after soaking process. The hides and skins are treated with fresh lime. In this process, nonstructural proteins, natural grease and fats are removed. The fibres are opened up. The fibres are swelled up to the desired extent. The hairs and other keratinous matters are removed. Some interfibrillary soluble proteins are also removed in this process. 2-3 days are taken to complete the liming operation. The pH is kept between 12-12.5.
- Unhairing: It is often linked with liming process. It is said that, unhairing is one of the most important objectives of liming process. The hairs are removed from the pelt in this stage. Lime and sulphide aids in unhairing process. It can be done by destroying or modifying the epidermis tissues or by attacking the hair itself. The hair is loosened more quickly in an old lime than in a fresh lime liquor. But it is not suggested because the swelling and plumping of the fibres can not be achieved using an old lime liquor.
- Fleshing: Fleshing is done after the liming process. It is done to remove the unfibrilliary proteins from the hides/skins.
- Splitting: The swelled and plumped hides or skins are cut into two or more layers according to the required thickness and type of leathers from the buyers.
- Deliming: The unhaired and fleshed hides and skins are taken for the next process which is deliming. In this process, the lime and other alkalis are removed from the limed pelt. By this process, the limed pelts are delimed. The non-structural proteins are also removed by this process. The swelling and plumping of leather are reduced. 1-2 hours of deliming is enough to achieve these objectives. The pH is lowered to a range between 8.2-8.5.
- Bating: It’s a generic term which refers to the use of proteolytic enzymes in leather manufacturing operations. The principal objectives of bating process are to produce the end products soft, stretchy, pliable and water vapor permeable. A silky grain surface is created in this process. It removes most of the alkaline materials from the hides and skins and creates porosity in it which improves its water vapor permeability. It removes plumpness of hides and skins by maintaining a pH range of 8.0-9.0.
- Scudding: It is done after bating process. Its main objectives are to remove the hair roots, dirt, natural pigments etc. During unhairing process, it can’t be done because of the swelling of pelt.
- Degreasing: It means removing grease from the hides or skins. Hides and skins contain high amount of natural grease. Natural fats or oils are stripped out from the hides or skins as much as possible. Such greases are responsible for uneven dying and finishing.
- Bleaching: It is done to yield a lighter colored pelt by chemically modifying the dark pigments.
- Pickling: It is the treatment process of the bated hides or skins with acid and salts solution. In this process the pH value is lowered to the acidic region to facilitate the chrome tanning process. It is done in presents of salt which is used to reduce acid swelling. The enzymatic action is stopped and astringency of chrome tanning agents are reduced by this process. Pickling helps in the penetration of chemicals like- chromium in the tanning process. The pH is brought down to 2.6-2.9 to make the hides or skins suitable for chrome tanning.
- Depickling: Rising of the pH to help the penetration of tanning agents. It is done when necessary.
- Tanning: Tanning is the method that transform the collagen protein of the raw hides or skins into a material which is not putrescible.
2.5. Crusting
- Sammying: The water is squeezed out from the leather (45-55%). It makes the leather hard and non slippery.
- Splitting: The hides or skins are splitted into several layers by splitting machine. It is done by a band knife which has an endless double beveled knife. The corium major portion of the leather has been cut to produce split or other types of leathers.
- Shaving: The leather is shaved to a get uniformity in thickness. The flesh side of the leather is forced through the sharp revolving knife. The flesh side is moved in the machine bed by pressing a treadle. The machine cuts the fibres off. The sharp blade of the shaving machine cut the leather in a proper angle.
- Wet back: The semi processed leather is being rehydrated to bring the weight of hides and skins back for crust operation.
- Rechroming: It gives fullness to the leather. It increases tensile strength, shrinkage temperature and softness of the hides and skins. pH is maintained in between 2.8-2.9
- Neutralisation: It is a deacidification process. It is done before dyeing, re-tanning and fatliquoring process. If it is not done prior to these operations, the dying materials will not spread uniformly over the leather surface. As a result, an uneven dying will be produced. It happens because, the surface will be acidic and the positive charges in the leather will not allow other chemicals to enter the interior portion of the leather. This process is very important for chrome and other mineral tanned leathers. In this process, the pH of the hide and skin is increased to the neutral pH. The leather surface of the neutralized leather is electrically neutral. It creates a proper environment for anionic chemicals. The pH is maintained in between 5.0-6.0 for chrome tanning.
- Re-tanning: The chrome tanned leather or aluminum-chrome tanned leathers are retanned to modify the physical properties of the finished leather. Additional tanning agents are added to improve the softness, fullness and grain tightness properties of the leather.
- Dyeing: Nearly all the leathers are dyed to enhance the appearance or look of the finished leather and make them fashionable in varying styles. Various colors are imparted to the leather as per the requirements from the buyer. Proper dyeing is necessary to make the leather acceptable in the ongoing market. It also improves its quality. It was practiced from prehistoric days. The dye gets fixed into the leather.
- Fatliquoring: It is done to coat the surface of fibres of the leather with fats and oils and to fix it there. If fatliquoring is not done, the leather will be hard and bony. The leather will be hard on drying and the fibres will break down. But, treating the leather with oils and fats will prevent its fibres from sticking together at the time of drying process. The total amounts of fats and oils must be adjusted first to assure the properties of the finished leather. It increases flexibility, softness and additional strength properties of the fibres.
- Filling: In this process, heavy chemicals are added to give the leather its desired fullness, roundness and plumpness. These materials penetrate deep into the leather and fill up all the voids in it specially in the looser fiber structures. They prevent those voided areas from being collapsed. Thus, it also facilitates in finishing, plating and in uniform buffing.
- Stuffing: fats or oils and waxes are added between the fibres.
- Whitening: The color of the leather is lightened by this process.
- Post Tanning Operations:
- Fixation: All unbound chemicals are chemically bonded or removed from leather in this process.
- Setting: By this operation, the surface area/grain are flattened and excess water from leather are removed. The growth marks in leather are also removed to some extent.
- Vacuum Drying: This process reduces the drying time. The leather is dried under fixed temperature and pressure. It depends on some factors like the thickness of the leather, drying temperature etc. It removes water from the leather and makes it soft.
- Hang to dry: It is then hanged for drying again for some days.
- Tunnel: It is a process of drying the leather with vapor. It makes the leather soft and smooth.
- Staking: The leather is being vibrated by staking machine which makes the leather more soft and smoother.
- Toggling: The surface area is prolonged as well as softened by this process.
- Trimming: The unwanted portion of leather is removed by this process.
- Plating: The leather is softened by heat treatment and its thickness is reduced.
2.6. Finishing
- Objectives of Finishing:
- It provides levelness of colour on the leather.
- It gives uniformity in shades from leather to leather.
- It provides the desired colour.
- A matt to gloss surface.
- It improves the properties of leather.
- It hides all the defects of the leather surface.
- It prevents mould or fungal growth.
- It makes the leather fast to light, acid, alkali etc.
- Embossing
- Ironing
- Curtain coating
- Glazing
- Padding
- Brushing
- Spraying
- Buffing
- Plating
- Impregnation
- Roller coating
- Opaque
2.7. Chemicals Used in the Leather Production
| Wetting agent |
| Sodium carbonate |
| Bactericide |
| Degreasing agent |
| Soda ash |
| Bactericide |
| Sodium chloride |
| Soaking enzyme |
| Lime |
| Sodium sulfide |
| Liming auxiliary |
| Polyphosphate |
| Degreasing agent |
| Polyphosphate |
| Ammonium sulfate |
| Deliming agent |
| Degreasing agent |
| Sodium meta bisulphite |
| Alkali |
| Bating Enzyme |
| Acidic Bating Enzyme |
| Degreasing agent |
| Salt |
| Sodium chlorite |
| Formic acid |
| Sulfuric acid |
| Hypo |
| Basic chromium sulfate |
| Sodium formate |
| Electrolyte stable fat/chrome stable fat |
| Fungicide |
| Wetting agent |
| Formic acid/ Acetic Acid |
| Chrome powder |
| Chrome syntan |
| Sodium formate |
| Remsol OCS |
| Relugan GT-50 |
| Sodium Bi-carbonate |
| Trisul ML/1C |
| Sodium formate |
| Neutralizing syntan |
| Sodium Bi-carbonate |
| Remsol C-2 |
| Mimosa |
| Quebracho |
| Gambier |
| Tara powder |
| Chest nut powder |
| Replacement syntan |
| Amphoteric syntan |
| Milling syntan |
| Melamine resin |
| Acrylic resin |
| Di-cyanine di-amide |
| Dispersing syntan |
| Dye |
| Dye leveler and penetrator |
| Semisynthetic fat |
| Synthetic fat |
| Raw oil |
| Fish oil |
| Lecithin fat |
| Waterproof fat |
| Polymeric fat |
| Phosphoric ester |
| Phospholipid fat |
| Electrolyte stable fat |
| Neat foot oil |
| Basic dye |
| Acid dye |
| Cationic fat |
| Pigment |
| Dye liquid |
| Wax TRA |
| Sensol SW |
| Clear Emulsion G |
| Encryl GC |
| Encryl XE |
| Ultra Filler |
| Wax FF |
| Penetrator |
| Silicon |
| NC Lacquers |
| Protein Binder |
| Acrylic Resin Binder |
| Polyurethane Binder |
Chapter Three
- Hazardous Chemicals and Their Risk Assessments

3.1. General Discussion
- Inhalation
- Skin contact
- Digestive system
- Injection
- Chemical hazard
- Biological hazard
- Physical hazard
- Psychological hazard
- sulfide
- Fatty acid
- Solvents
- Lacquers
- Formaldehyde
- Old Lime liquor containing ammonia and sodium sulfide.
3.2. Pictograph of Hazardous Chemicals
- Toxic
- Health Hazard
- Corrosive
- Oxidizing
- Flammable
- Explosive
- Irritant
- Environmental Hazard etc.

3.3. Highly Potential Hazard Group
- Sulfuric acid
- Acetic acid
- Formic acid
- Ammonia
- Hydrogen Peroxide
- Sodium Hydroxide
- Formaldehyde
- Sulphides and Hydrosulphides
- Lime
- Glutaraldehyde
- Sodium Chlorite
- Molecular weight: 98.0
- Area of use: Pickling
- Reactivity: It is highly reactive and corrosive. Spattering occurs if water is added to acid.
- It burns the skin when come in contact with skin.
- It causes irritation.
- Coughing.
- It makes breathing difficult.
- Molecular weight: 60.05
- Area of use: Acid wash, Dyeing, Fatliquoring
- Hazard Data: It is highly flammable, highly reactive.
- Acetic acid vapor cause irritation of the eyes, nose and lungs.
- It causes nausea
- It can also cause diarrhoea
- If comes in contact with the skin, it will be able to burn it.
- Molecular weight: 46.0
- Area of use: Acid wash, Dyeing, Re-chroming, Fatliquoring.
- Burns the skin if come in contact with it.
- Irritation
- Headache
- Dizziness
- It can also cause nausea.
- Molecular weight: 17.0
- Area of use: Deliming
- Hazard data: It is flammable and explosive when its concentration level is in between 16-25%.
- It causes skin irritation
- Skin burning is another risk that carries with it.
- It makes breathing very difficult.
- It also causes chest pain.
- Molecular weight: 34.0
- Area of use: Unhairing
- Hazard Data: It is highly reactive and form explosive compounds if reacts with organic materials, reducing agent.
- It causes whitening of skin
- Discomforts in eye and nose
- Irritation
- Burning of skin
- Molecular weight: 40.0
- Area of use: Liming, Reliming
- Hazard Data: They are highly reactive. They produce flammable, explosive H2S gas.
- They cause respiratory irritation
- They can burn the skin
- Vomiting
- Molecular weight: 30.0
- Hazard Data: It is highly flammable. It generates toxic vapor. Maximum allowed 1.2 mg/L
- Vomiting
- Headache
- Irritation of eyes, skins
- It can cause even collapse.
- Molecular weight: 34.0
- Area of use: Liming, Unhairing agent
- Hazard Data: It is highly flammable.
- Irritations of eyes and nose at high concern.
- It can paralyse the respiratory system.
- Molecular weight: 100.0
- Area of use: Fatliquoring
- Hazard data: It is flammable.
- It causes nausea
- Irritations of skin and eyes
- Coughing
- Molecular weight: 90.0
- Area of use: Pickling
- Hazzard Data: It is flammable.
- Respiratory systems problem
- Skin problem
3.4. Moderate Potential Hazardous Group
- 01
- Aluminium sulfate
- 02
- Amyl acetate
- 03
- Amyl alcohol
- 04
- Benzyl alcohol
- 05
- Chromium salts
- 06
- Isopropyl alcohol
- 07
- White spirit
3.5. Low Potential Hazard Group
- 01.
- Alum oils
- 02.
- Acetone
- 03.
- Paraffin
- 04.
- Borax
- 05.
- Boric acid
- 06.
- Pigment
- 07.
- Albumen
- 08.
- Ammonium chloride
- 09.
- Sodium acetate
- 10.
- Casein
- 11.
- Sodium bicarbonate
- 12.
- Calcium chloride
- 13.
- Lactic acid
- 14.
- Titanium salts
- 15.
- Sodium nitrite
- 16.
- Ferrous acetate
- 17.
- Sodium phthalate
3.6. Miscellaneous Hazard Group
Chapter Four
- Leather Industry and Their Effect on Environment

4.1. Environment
4.2. Pollution
4.3. Pollutant
- Concentration
- Persistence
4.4. Sources and Nature of Pollutant in Leather Industry
- Raw skin trimmings
- Fleshing wastes
- Wet blue splits
- Shaving dust
- Buffing dust
- Leather trimmings
4.5. Characteristics of Tannery Discharge
- Continuous flow wastes
- Intermittent flow wastes
4.6. Effects of Hazardous Chemicals in Water
- The chromium and sulphide are toxic to the microorganisms and they disturb the biological treatment operation.
- The suspended lime also interferes in the biological activities.
- The dissolved chromium produces toxicity that is dangerous for fish and aquatic life.
- Acidic effluent from pickling process is corrosive. They can damage the concrete or metal pipes.
- Lead, cadmium produce carcinogenic effect which is responsible for cancer.
- Formaldehyde is also responsible for this. The use of formaldehyde is unavoidable in the leather manufacturing process. This is because it acts as a cross linker for casein top coats. [Ram Mohan-2008]
4.7. Central Effluent Treatment Plant

4.8. Environmental Impact of the Leather Industry
- Ammonia
- Sulphides
- Formaldehydes
- Chlorine gas
- Fatty acids

Chapter Five
- Result and Discussion

5.1. Effect Description on the Basis of Interview
-
Do they feel vomiting while working in the tannery?
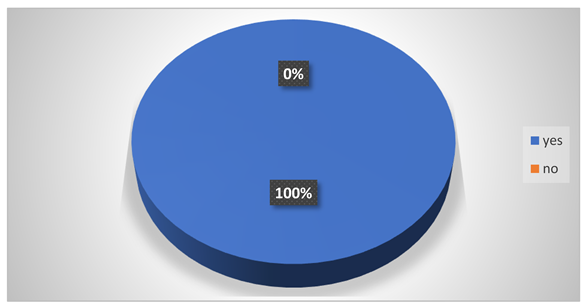
-
Do they feel any hearing problem?
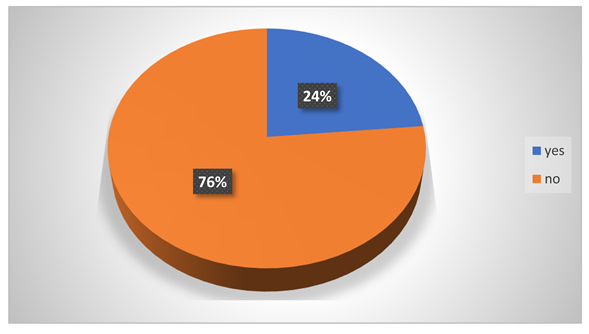
-
Do they have reduced eyesight?
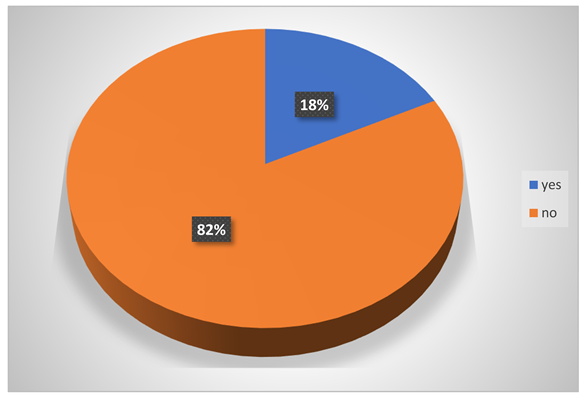
-
Do they have any breathing problem?
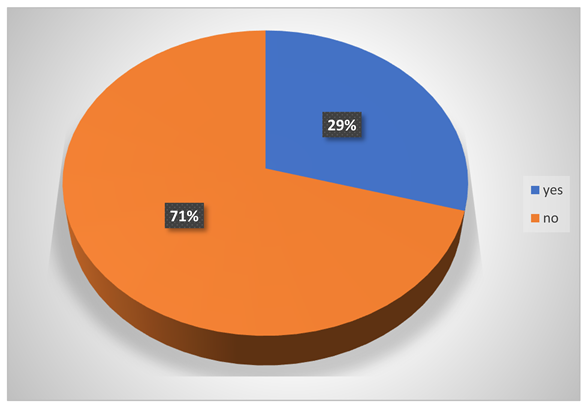
-
Do they have hair fall problem?
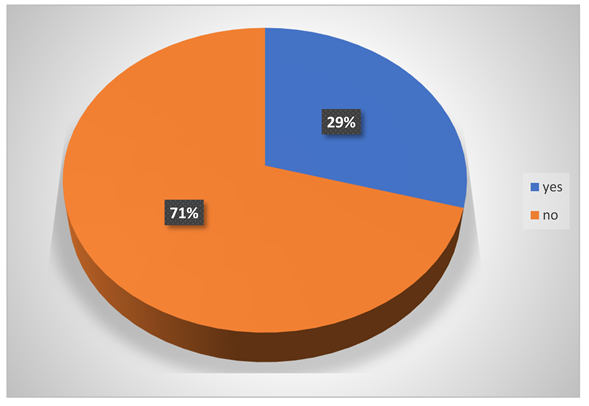
-
Do they have liver problems?
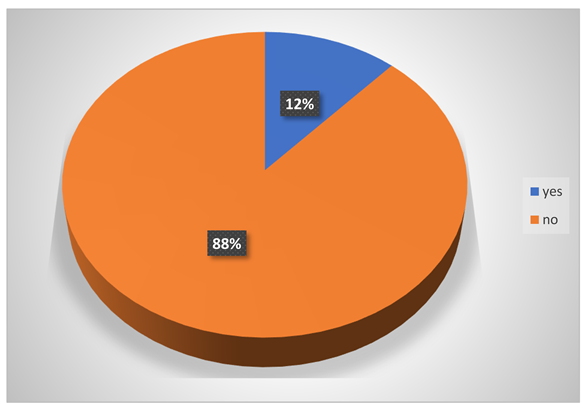
-
Does eating food in the middle of work cause stomach problems to them?
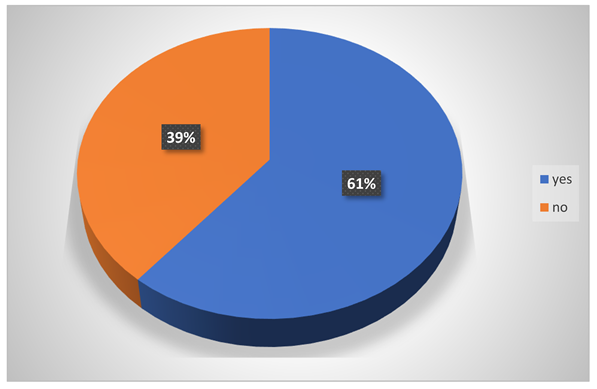
-
Do they have sound sleep?
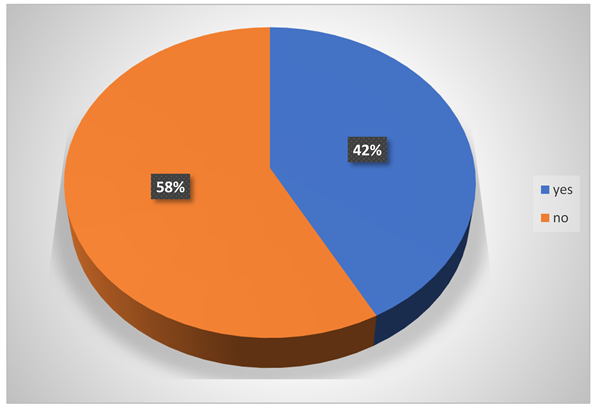
-
Did they ever affect by acid?
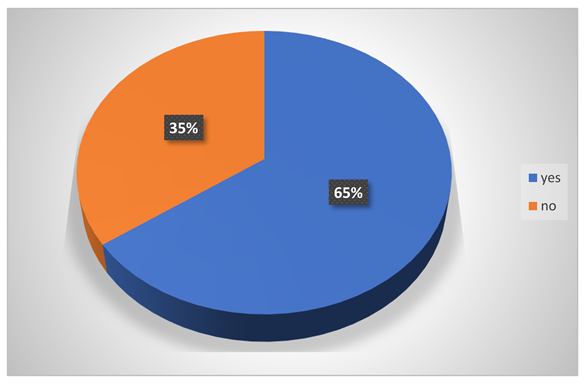
-
Did they have any eye contact with chemicals?
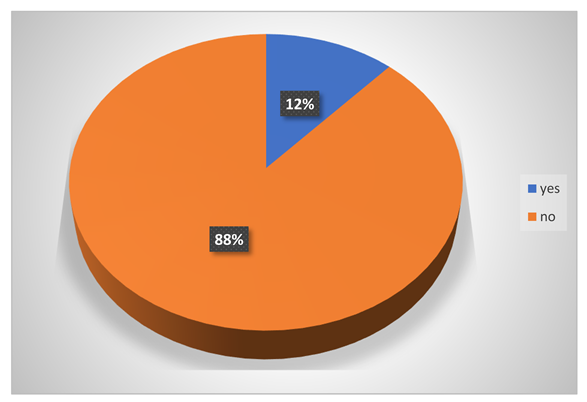
-
Did any chemicals cause wounds in their body?
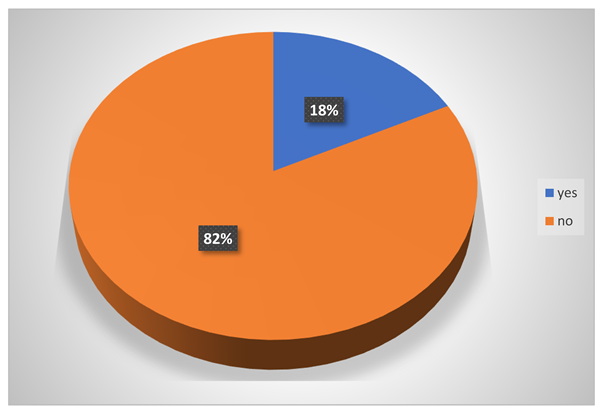
-
Do they have allergy problem?
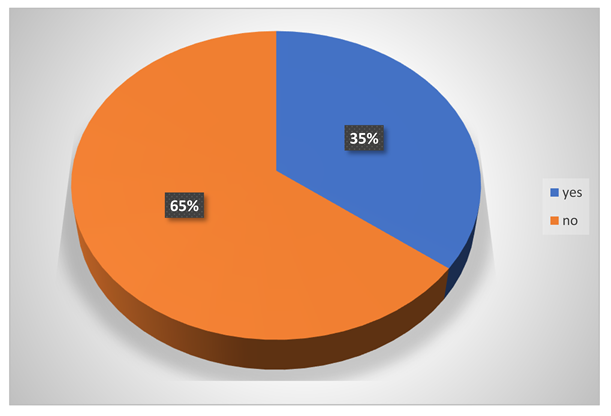
-
Do they get adequate protection during handling hazardous chemicals?
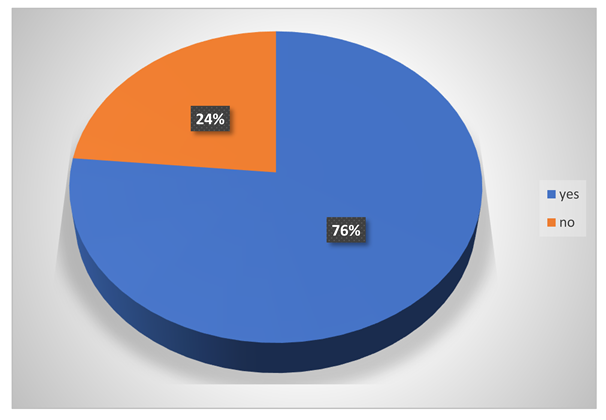
-
Do the have any ideas about the effects of harmful chemicals?
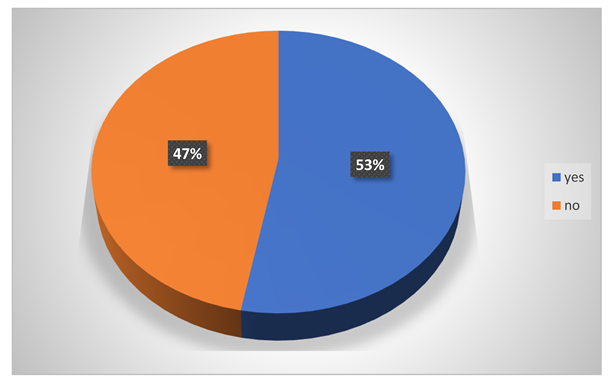
-
Do they have any problem of frequent diarrhoea?
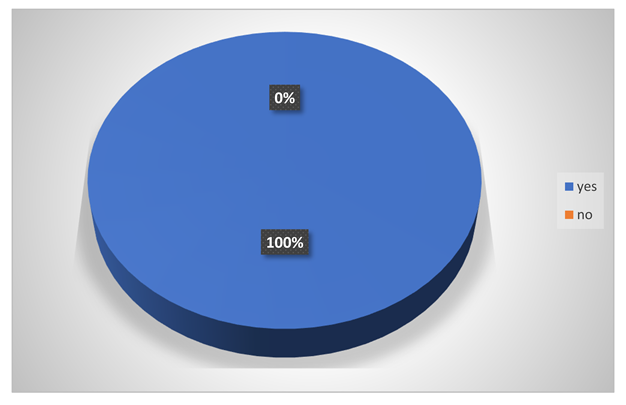
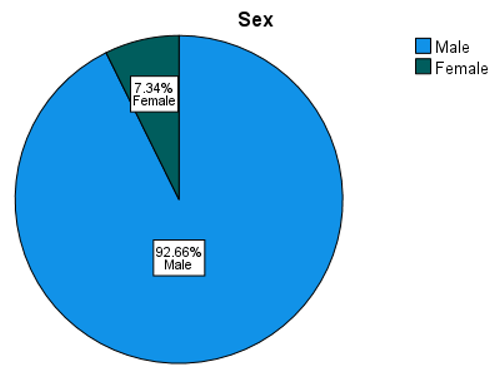
5.2. Univariate Analysis
| Sex | ||
| N | % | |
| Male | 303 | 92.7% |
| Female | 24 | 7.3% |
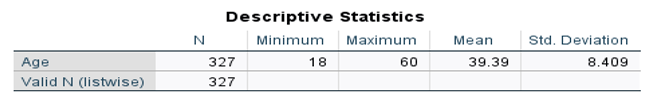
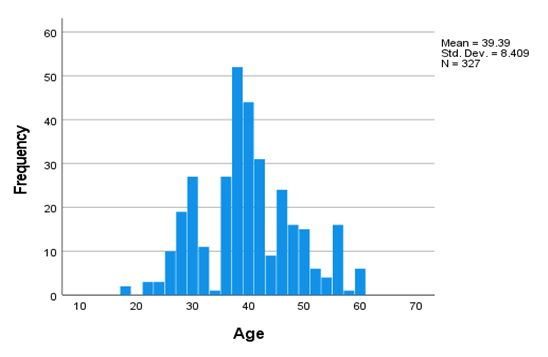
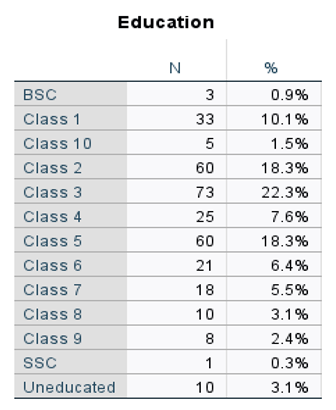
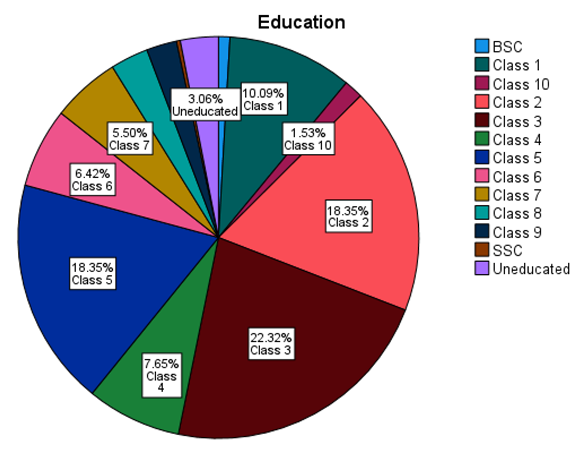
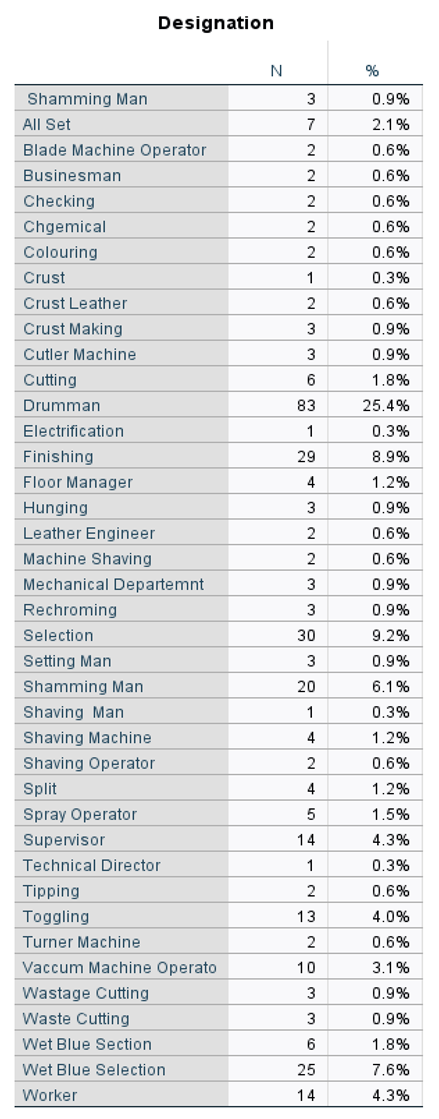
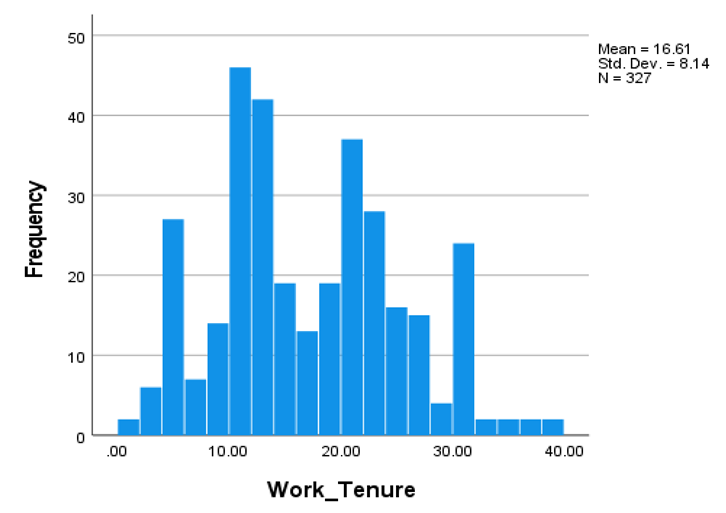
5.3. Affected People
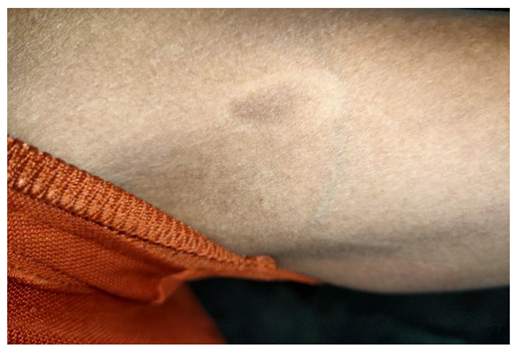
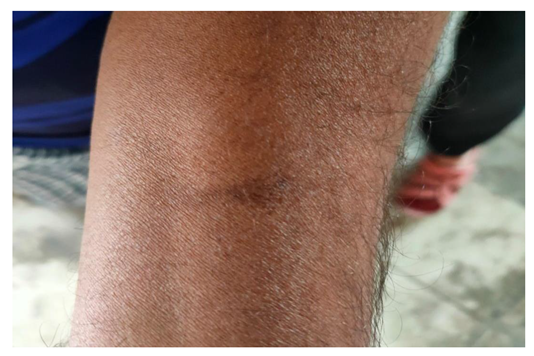
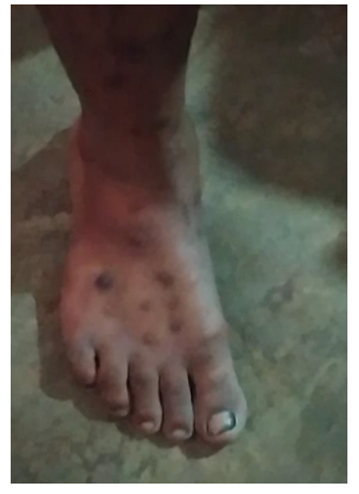
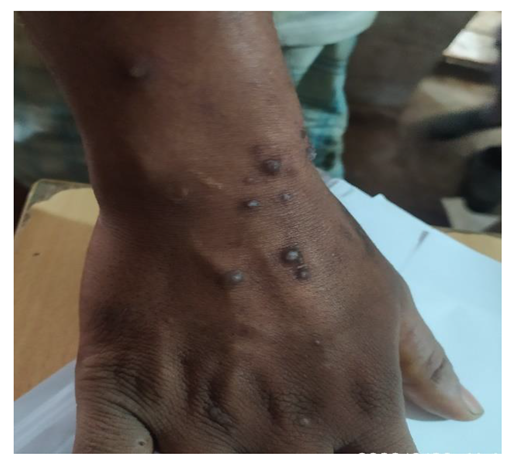
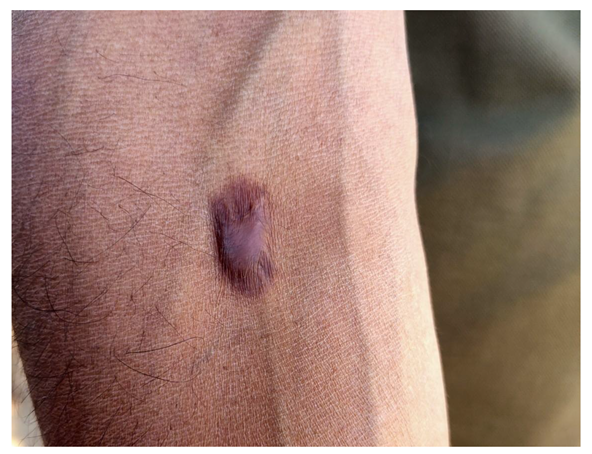
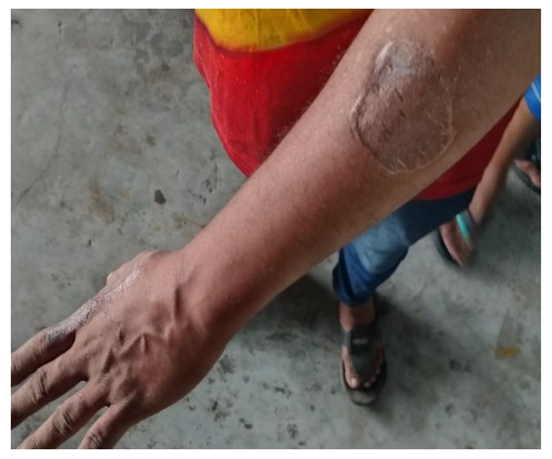
5.4. Impact on Labor Efficiency
5.5. Instructions for Storing Hazardous and Other Chemicals
- Combustible
- Corrosive
- Flammable
- Irritating
- Toxic
5.6. Personal Protective Equipment
- Hand gloves should be checked every time for leakage before using it. A leak hand glove is dangerous for hand. Thick and long hand gloves are preferred as there is more protection.
- Googles must be used while pouring liquid chemicals to protect the eyes.
- Safety shoes must withstand attack by any hazardous, toxic or corrosive chemicals specially against acid, alkalis etc.
- Masks must be weared while working with chemicals which can produce noxious smells.

5.7. Work Place Criteria
- The working floor should be kept neat and clean.
- Slip materials should be kept away from the wet floor area. Safety shoes must be weared by the workers of this area.
- The drain must be covered otherwise there will be accidents frequently.
- The drainage system must be free of clutter and also good enough to carry the liquid wastes quickly from the processing area.
- Shaving dust, fleshing dust, trimming dust must be cleaned properly from the working floor.
- Frequent checking for the spillage of chemicals is necessary.
Chapter – 6
- Safety Compliance of Leather Industry
6.1. Compliance
6.1.1. Dust and Fumes

6.1.2. Ventilation Temperature
6.1.3. Place for Children

6.1.4. Overcrowding
6.1.5. Sound
6.1.6. Health Care
6.1.7. First Aid
6.1.8. Safety Record Book
6.1.9. Shelters

6.2. Maternity Benefit
-
The employer has reason to believe, Removal
- Demotion to a lower post, grade or scale of pay for a period not exceeding one year.
- Remission of promotion for a period not exceeding one year.
- Withholding of increment for a period of not exceeding one year fine.
- Censure or warning.
6.3. Worker’s Responsibility
- Workers should give their best for increasing productivity.
- Workers should count money at the time of receiving wages.
- Should inform immediately to the responsible person if any mistake found in wage calculation.
- Should not be absent without granted leave.
- Should give 60days notice before resigning from service.
- Should apply for leave in advance.
- Report immediately to the superior if not being able to attend the factory due to any unavoidable circumstances, such as sudden sickness, accident or else.
- Workers may get less wages than they deserve due to non-compliance in wages and benefit payments.
- Delayed payment of wage, Non-compliances in termination practices, granting leave, and tile handling of misconducts may create dis-satisfaction among workers.
- Loss of worker’s motivation.
- Increase of worker migration rate.
- Risk of losing business from buyers.
6.4. Maximum Working Hours
6.5. Use of PPE

6.6. Ergonomics
- The study of individual operator or working team.
- The provision of data design.
6.7. Conclusion
- Identification: This is the identification of products, in short chemical identity.
- Hazardous identification: whether it is hazardous or not.
- The composition and information on ingredients.
- There should be stated about fire fighting measures
- The first aid measurements should be included.
- The process of accidental release measurements
- Handling and storage. It must include the way of safe use of chemicals.
- Exposure controls and personal protection measures for these chemicals.
- Chemical and physical properties of the chemicals.
- Stability and reactivity of the chemicals.
- Ecological information of the chemicals is also necessary.
- Toxicological information is a must.
- The process of disposing process
- Regulatory information can be added.
- Transport informations may also be added.
6.8. Recommendation
- The management must educate the workers about all the chemicals and their negative effects.
- The industry must provide the workers their personal protective equipment and also force them to wear it during work to avoid unwanted accidents.
- Accidents can happen anytime in the industry. So, here must have first aid service in the industry.
- Instant ambulance service is also recommendable for serious injuries.
- The continuing of salary is recommended. The workers always fear that, if they get sickened and can’t go to the industry; they will lose their job. This fear should be driven away from their mind.
- All the chemicals should be arranged and stored according to their hazard level. It will reduce the accident rate in the tannery.
- The drainage system should be covered properly.
6.9. Discussion
Acknowledgments
References
- Akhi Akter, Mir Abdullah Al Mahfuz; An overview of Bangladesh Leather Industry, December 17,2018.
- “An Introduction to the Principles of Leather Manufacture”-Sasanka Sekhar Dutta.
- ATSDR, 2008. Toxicological Profile for Cadmium. Agency for Toxic Substances and Disease Register.
- “Bangladesh: Tanneries Harm on Workers, Poison Communities.” -Human Right Watch.
- Department of Environment, Ministry of Environment and Forest, Bangladesh.
- Environmental Commission of L.U.L.T.C.S: IUE Recommendation on Cleaner Technology for Leather Production. Updated document, London, 1997.
- Hagerman, A.E. 1980 Ph. D. Thesis, Purdue University, West Lafayette, IN, USA.
- Han, B. Jaurequi, J. Tang. B. W., Nimni, M. E., 2001. Proanthocyanidin: A Natural Cross-Linking Reagent for Stabilizing Collagen Matrices. J. Biomed Mater. Res. Part A 65,118-124. [CrossRef]
- Hasnat A. Rahman, Pasha M (2013), Assessment of Environmental Impact for Tannery Industries in Bangladesh. Int. J Environmental Science, Dev 4(2):217-220. [CrossRef]
- IARC., 2004. Monographs on the Evaluation of Carcinogenic Risks to Humans. In: Inorganic & Organic Lead Compounds Vol. 87. International Agency for Research on Cancer, pp. 10-17 [LID 7420].
- Louis, E.D., Jurewicz, E.C., Applegate, L.K., V. Graziano, J.H., Carroll, S. Todd, A. 2003. Association Between Essential Tremor & Blood Lead Concentration. Environ. Health Penspect, 111, 1707-1711. [CrossRef]
- Rajamani S: Appropriate Chrome Recovery and Reuse System -Experience in Indian Tanneries. Proceedings of the Workshop on Pollution Abatement and Waste Management in the Tanning Industry for Countries of the Danube River Basin, Ljubljana, 1995.
- Ram Mohan, C, Saravanabhava, S. Thanikaivelan, P. Raghava Rao, J. Nair, B. U, 2008.Development of Formaldehyde Free Retanning Syntans, Clean Technol. Environ policy 10,287-294. [CrossRef]
- Safety Manual on Leather Processing, Central Leather Research Institute, Adyar, Chennai 600 200, 1999.
- Sumita Dixit, Ashish Yadav, Premendra D. Dwivedi, Mukul Das et al. 2014: Toxic Hazards of Leather Industry and Technologies to Combat Threat: A Review. Journal of Cleaner Production 87(2015) 39-49. [CrossRef]
- Suresh, V. Kanthimathi, M. Thanikaivelan, P. Rao Raghva, J. Nair, B.U, 2001. An Improved Production Process for Cleaner Chrome Tanning in Leather Processing. J. Clean prod.9,483-491. [CrossRef]
- Theory and Practice of Leather Manufacture. -by K. T. Sarkar.
- The Daily Jugantor, 24th September, 2021.
- The Daily Star, 13th September, 2021.
- The Dhaka Tribune, 1st October, 2021.
- Internet /Wikipedia.
- Putri, D.O., et. al. (2017). The effects of occupational health and safety, work environment and discipline on employee performance in a consumer goods company. IOP Publishing. [CrossRef]
- International Labor standards on occupational safety and health: working environment (Air pollution, noise and vibration) convention,1977(No.148) International labor organization.
- International labor standards on working time: Hours of works (commerce and offices) convention,1930 (no.30). International labor organization.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



